Introduction
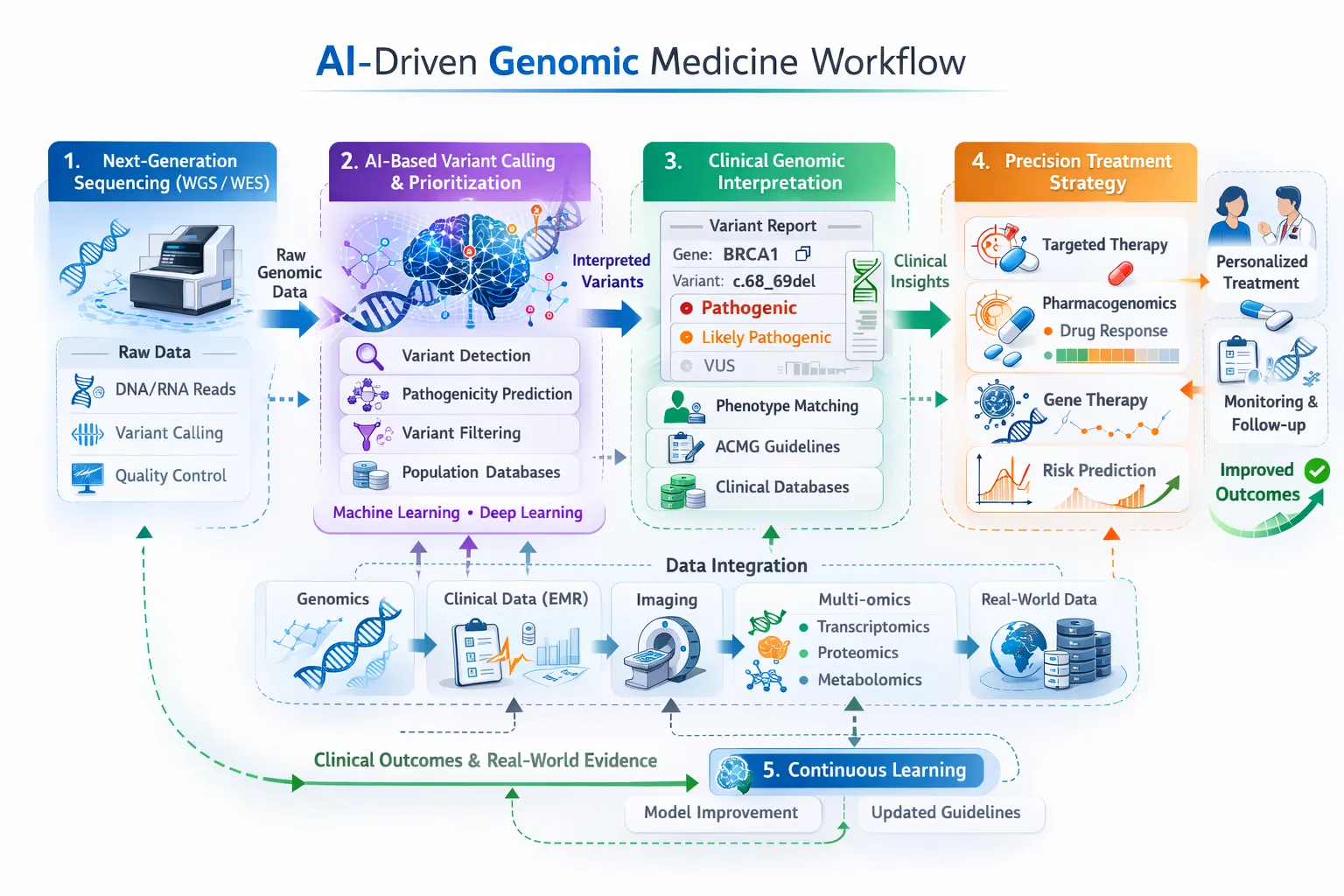
Advances in DNA sequencing technologies have made it possible to generate enormous amounts of genomic data from individual patients. A single whole-genome sequence contains roughly 3 billion DNA base pairs and millions of genetic variants. Interpreting this information to identify clinically meaningful insights presents a major challenge for researchers and clinicians.
Artificial intelligence (AI) is increasingly being used to address this challenge. By applying machine learning and data-driven computational approaches, AI systems can analyze large genomic datasets, identify patterns associated with disease, and support clinical decision-making. As a result, AI is emerging as a key component of genomic medicine and precision healthcare.
Why AI Is Needed in Genomic Medicine
Modern genomic technologies such as whole-genome sequencing (WGS) and whole-exome sequencing (WES) generate vast quantities of data. A typical human genome contains:
- ~3 billion DNA base pairs
- ~4–5 million genetic variants compared with the reference genome
- thousands of variants of uncertain significance
Only a small proportion of these variants contribute to disease. Identifying clinically relevant mutations therefore requires complex computational analysis.
AI and machine learning tools can help:
- prioritize disease-causing variants
- identify patterns across large genomic datasets
- predict functional effects of mutations
- integrate genomic data with clinical information
These capabilities make AI particularly valuable in clinical genomics and precision medicine.
Key Applications of AI in Genomic Medicine
Variant Interpretation
One of the most important applications of AI in genomic medicine is predicting whether genetic variants are harmful or benign.
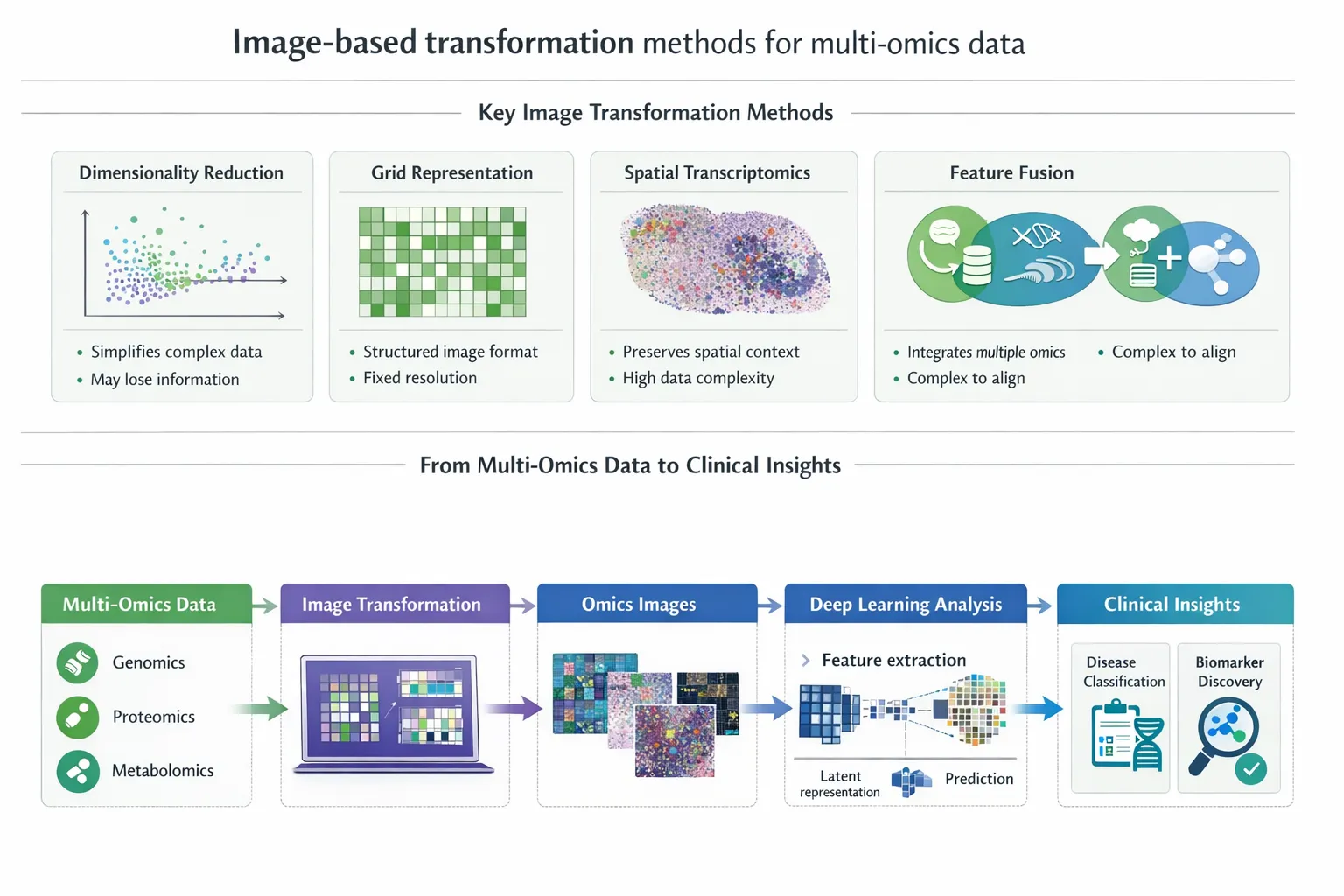
Machine learning models can analyze multiple types of data simultaneously, including:
- DNA sequence context
- evolutionary conservation
- protein structure
- gene expression patterns
Tools such as DeepVariant and AlphaMissense use deep learning to improve variant calling and pathogenicity prediction.
For example, deep neural networks have been shown to improve the accuracy of identifying genetic variants from sequencing data, helping clinicians detect disease-associated mutations more reliably (Poplin et al., 2018).
Rare Disease Diagnosis
Genomic sequencing has become an important diagnostic tool for patients with rare or undiagnosed diseases. However, identifying the causal mutation among millions of variants is challenging.
AI-based clinical genomics platforms can combine genomic data with patient symptoms and clinical records to prioritize likely disease-causing variants.
For instance, computational systems such as Exomiser integrate:
- genomic sequencing data
- phenotype descriptions
- known disease-gene associations
to help clinicians identify candidate variants responsible for rare genetic disorders (Robinson et al., 2014).
Studies suggest that genomic sequencing combined with advanced computational analysis can provide diagnoses for 25–40% of patients with previously unexplained rare diseases (Wright et al., 2018).
Cancer Genomics and Precision Oncology
Cancer is fundamentally a disease of genetic mutations. Tumour sequencing is therefore increasingly used to guide treatment decisions.
AI systems can analyze tumour genomic data to:
- identify driver mutations
- classify tumour subtypes
- predict treatment response
- discover new therapeutic targets
For example, machine learning algorithms can analyze large cancer genomics datasets such as The Cancer Genome Atlas (TCGA) to identify genomic patterns associated with tumour progression or drug sensitivity.
In clinical practice, genomic profiling of tumours can help match patients to targeted therapies designed to inhibit specific mutated proteins.
Drug Discovery and Development
AI is also transforming the process of discovering new medicines.
By analyzing genomic and biological data, AI systems can:
- identify disease-associated genes
- predict drug targets
- simulate drug–protein interactions
- accelerate clinical trial design
Machine learning approaches have already been used to identify potential therapeutic targets for conditions such as cancer, neurological diseases, and rare genetic disorders.
Integrating genomic data into drug development may also enable more precise therapies tailored to specific genetic profiles.
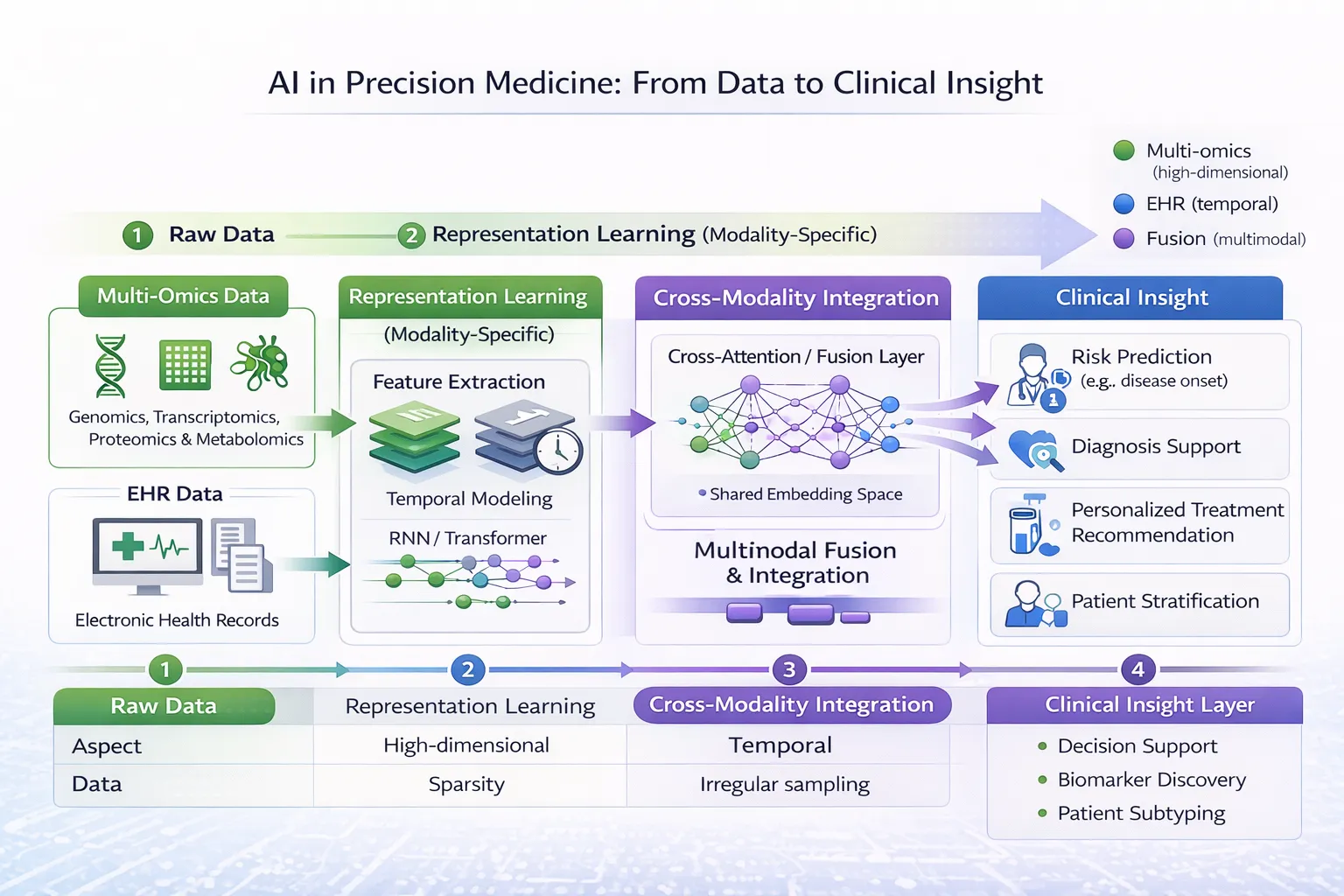
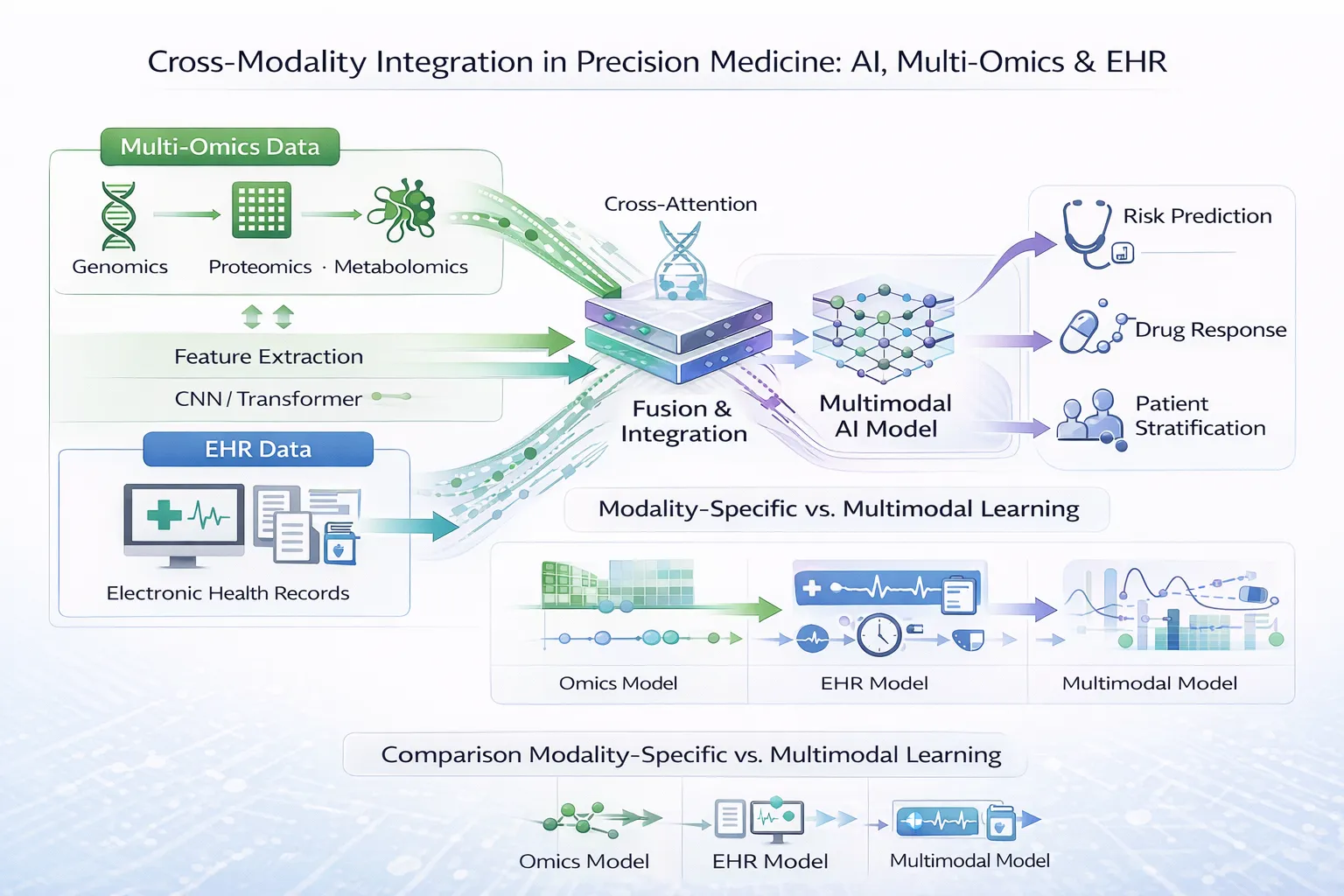
Integrating Genomics with Electronic Medical Records
A key goal of genomic medicine is integrating genetic data with routine clinical information stored in electronic medical records (EMRs).
AI can help combine multiple data sources, including:
- genomic sequencing data
- laboratory results
- imaging data
- clinical history
- lifestyle and environmental information
By analyzing these datasets together, AI systems may help clinicians identify disease risk, guide treatment decisions, and predict patient outcomes.
Such integrated approaches are central to the concept of precision medicine, which aims to tailor healthcare to the biological characteristics of each individual.
Challenges and Limitations
Despite its promise, the use of AI in genomic medicine faces several important challenges.
Data Quality and Bias
AI systems are only as reliable as the data used to train them. Many genomic databases currently overrepresent populations of European ancestry, which may limit the accuracy of predictive models for other populations.
Interpretation of AI Models
Some AI systems function as “black boxes,” making it difficult to understand how predictions are generated. Improving model transparency is important for clinical adoption.
Privacy and Ethical Considerations
Genomic data is highly sensitive personal information. Safeguarding patient privacy and ensuring responsible data use are essential when deploying AI systems in healthcare.
Regulatory Oversight
AI-driven diagnostic tools must undergo rigorous validation to ensure safety and effectiveness before being widely adopted in clinical settings.
The Future of AI in Genomic Medicine
As genomic sequencing becomes more common in healthcare, the role of AI will continue to expand. Future developments may include:
- AI-driven variant interpretation systems
- real-time genomic clinical decision support tools
- integrated multi-omics analysis platforms
- predictive models for disease risk and treatment response
By combining genomics, clinical data, and advanced computational methods, AI has the potential to transform medicine from a reactive system to a predictive and personalized healthcare model.
Conclusion
Artificial intelligence is rapidly becoming an essential tool in genomic medicine. By enabling the analysis of complex biological datasets, AI can help identify disease-causing mutations, improve diagnostic accuracy, guide targeted therapies, and accelerate drug discovery.
While challenges remain, the integration of AI with genomic technologies represents a major step toward the realization of precision medicine, where treatments and prevention strategies are tailored to the genetic and biological characteristics of each individual.