Introduction
Over the past decade, CRISPR gene editing has transformed the field of genetics. What began as a bacterial immune defense mechanism has rapidly evolved into one of the most powerful tools in modern biology. Today, CRISPR technology allows scientists to precisely modify DNA, offering the potential to treat genetic diseases at their source.
For many conditions that were once considered untreatable, gene editing raises the possibility of correcting disease-causing mutations rather than simply managing symptoms. As clinical trials expand and early therapies begin to reach patients, CRISPR is moving from the research laboratory—often described as “the bench”—into real-world medical treatments, or “the bedside.”
But how does CRISPR work, and what does its growing use mean for the future of medicine?
How CRISPR Works
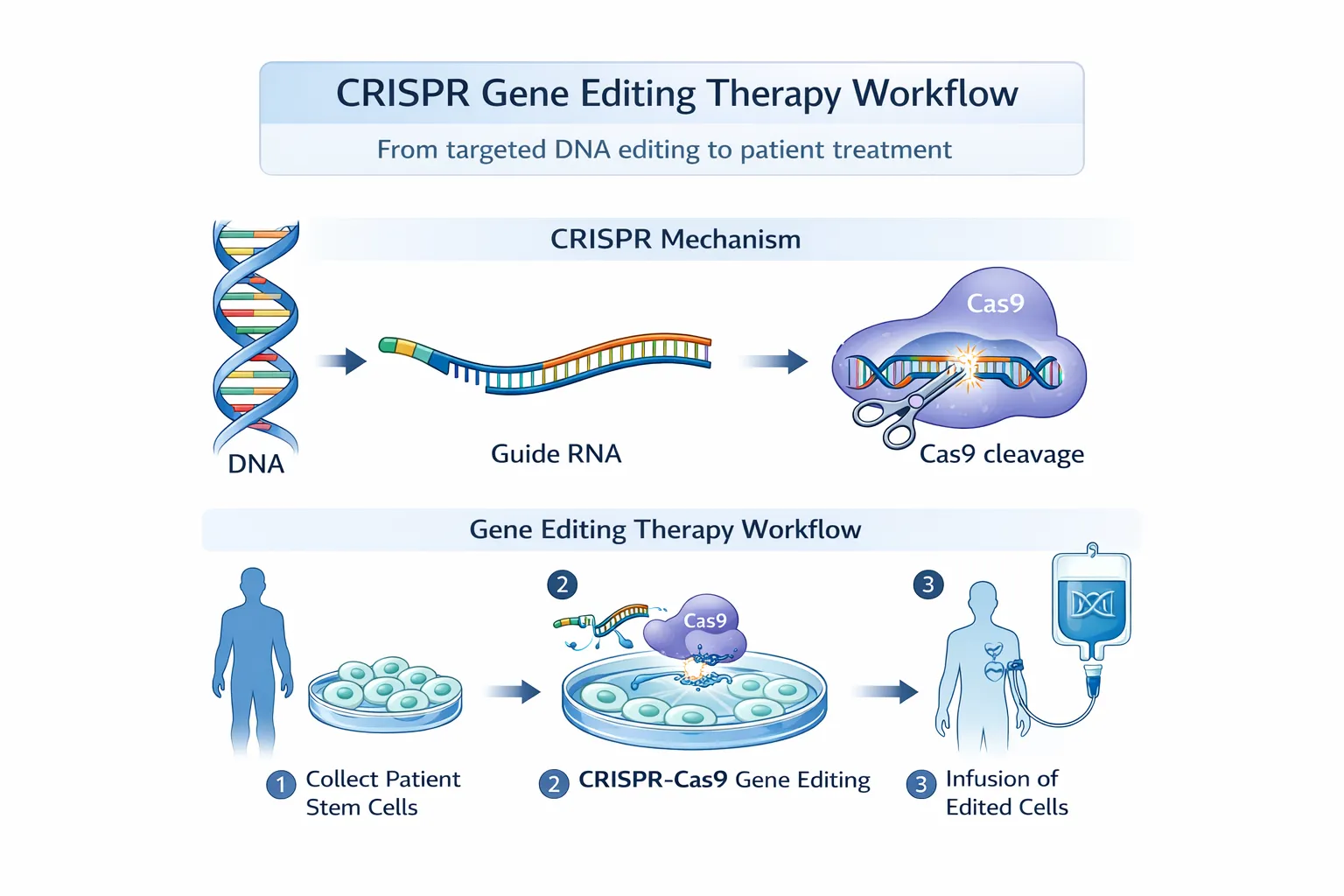
CRISPR stands for Clustered Regularly Interspaced Short Palindromic Repeats, a system originally discovered in bacteria. In nature, bacteria use CRISPR as part of their immune system to detect and destroy invading viruses.
Scientists adapted this system into a powerful gene-editing tool by combining two main components:
1. Guide RNA (gRNA) A short RNA sequence designed to match a specific region of DNA. This RNA acts like a molecular “address label,” guiding the editing machinery to the correct location in the genome.
2. Cas9 enzyme Cas9 is a protein that acts like molecular scissors. Once guided to the correct DNA sequence, Cas9 cuts the DNA strand.
After the DNA is cut, the cell attempts to repair the break. During this repair process, scientists can:
- Disable a gene by introducing small mutations
- Correct a faulty gene by inserting a healthy DNA sequence
- Insert new genetic material into the genome
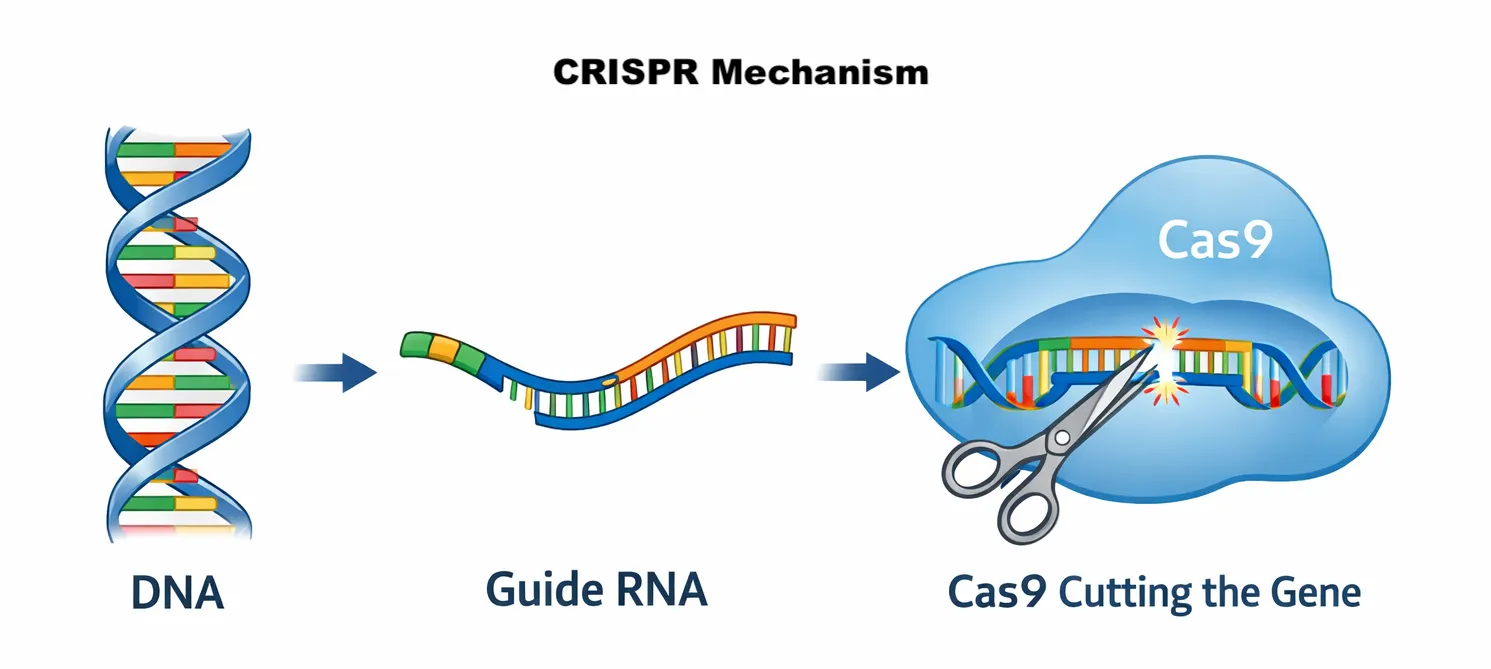
This ability to precisely edit DNA makes CRISPR far more efficient and flexible than earlier gene-editing technologies.
Clinical Applications of CRISPR
CRISPR technology is now being explored across a wide range of medical conditions, particularly diseases caused by genetic mutations.
Sickle Cell Disease
One of the most promising clinical applications of CRISPR is the treatment of sickle cell disease, a genetic blood disorder caused by a mutation in the hemoglobin gene.
In sickle cell disease, red blood cells become rigid and crescent-shaped, leading to:
- chronic pain
- anemia
- organ damage
- reduced life expectancy
CRISPR-based therapies aim to correct this problem by editing a patient’s own stem cells. The process typically involves:
- Collecting blood-forming stem cells from the patient
- Editing the cells using CRISPR to restore healthy hemoglobin production
- Reinfusing the corrected cells back into the patient
Once the edited stem cells begin producing healthy red blood cells, symptoms can be dramatically reduced.
Early clinical trials have shown encouraging results, with some patients experiencing long-term relief from severe symptoms.
Other Emerging Applications
Beyond sickle cell disease, CRISPR is being investigated for a growing number of conditions, including:
Genetic Eye Diseases
Inherited retinal disorders such as Leber congenital amaurosis are early targets for gene-editing therapies. Because the eye is relatively accessible and immune-privileged, it provides a promising setting for testing new gene-editing approaches.
Cancer Immunotherapy
CRISPR is also being used to engineer immune cells that better recognize and attack cancer. For example, researchers can edit T cells to enhance their ability to target tumor cells.
Rare Genetic Disorders
Many rare diseases are caused by mutations in a single gene. CRISPR offers a potential strategy for correcting these mutations directly.
Conditions currently under investigation include:
- muscular dystrophy
- metabolic disorders
- inherited immune deficiencies
Although many of these therapies remain in experimental stages, they demonstrate the broad potential of gene editing in medicine.
Safety Considerations
Despite its remarkable promise, CRISPR gene editing must overcome several important safety challenges before widespread clinical use.
Off-Target Effects
One of the main concerns is off-target editing, where the CRISPR system accidentally cuts DNA at unintended locations in the genome. Such unintended edits could potentially disrupt other genes or lead to harmful consequences.
Researchers are developing improved CRISPR systems with:
- higher precision targeting
- engineered Cas enzymes
- enhanced guide RNA design
These advances are helping reduce the likelihood of unintended edits.
Delivery Challenges
Another major challenge involves delivering CRISPR components to the correct cells inside the body.
Current delivery strategies include:
- viral vectors that carry gene-editing instructions
- lipid nanoparticles that transport CRISPR molecules
- direct editing of cells outside the body (ex vivo editing)
Each approach has advantages and limitations, and researchers continue to refine delivery technologies for different diseases.
Ethical Considerations
Gene editing raises important ethical questions, particularly when changes to DNA could be passed on to future generations.
Somatic vs Germline Editing
Most current CRISPR therapies involve somatic gene editing, meaning the DNA changes occur only in the treated individual’s cells and are not inherited by offspring.
This type of editing is generally considered ethically acceptable when used to treat disease.
In contrast, germline editing modifies sperm, eggs, or embryos, potentially affecting future generations. Because these changes are permanent and heritable, germline editing remains highly controversial and is tightly regulated or prohibited in many countries.
Equity and Access
Another ethical issue concerns fair access to advanced therapies. Gene-editing treatments can be extremely complex and expensive, raising concerns about whether these innovations will be available to all patients who need them.
Ensuring equitable access to gene therapies will be an important challenge as these treatments move into routine clinical practice.
The Future of Gene Editing
CRISPR technology continues to evolve rapidly. New techniques are being developed that allow even greater precision in editing DNA, including:
- base editing, which changes individual DNA letters without cutting the DNA strand
- prime editing, which enables highly precise genetic corrections
- improved delivery systems for targeting specific tissues
These advances could expand the range of diseases that gene editing can safely treat.
As research progresses, CRISPR is likely to play an increasingly important role in precision medicine, enabling treatments that directly address the genetic causes of disease.
Key Takeaway
CRISPR gene editing represents one of the most significant biomedical innovations of the 21st century. By enabling precise modifications to DNA, this technology offers new possibilities for treating genetic diseases that were once considered incurable.
While important scientific, ethical, and regulatory challenges remain, CRISPR is steadily moving from experimental research toward real-world medical therapies—bringing the promise of genetic medicine from bench to bedside.