Introduction
Modern medicine is undergoing a transformation. Advances in genomics, large-scale health data, and digital health records are enabling a shift from a “one-size-fits-all” model of healthcare to precision medicine—an approach that tailors prevention, diagnosis, and treatment to an individual’s biology and environment.
A recent research analysis published in Nature Communications outlines a roadmap for achieving this goal by integrating genomic data, electronic health records (EHRs), and multi-omics technologies. ([Nature][1])
This article explains how these systems work together and what they mean for patients, clinicians, and the future of healthcare.
The Post-Genomic Era of Medicine
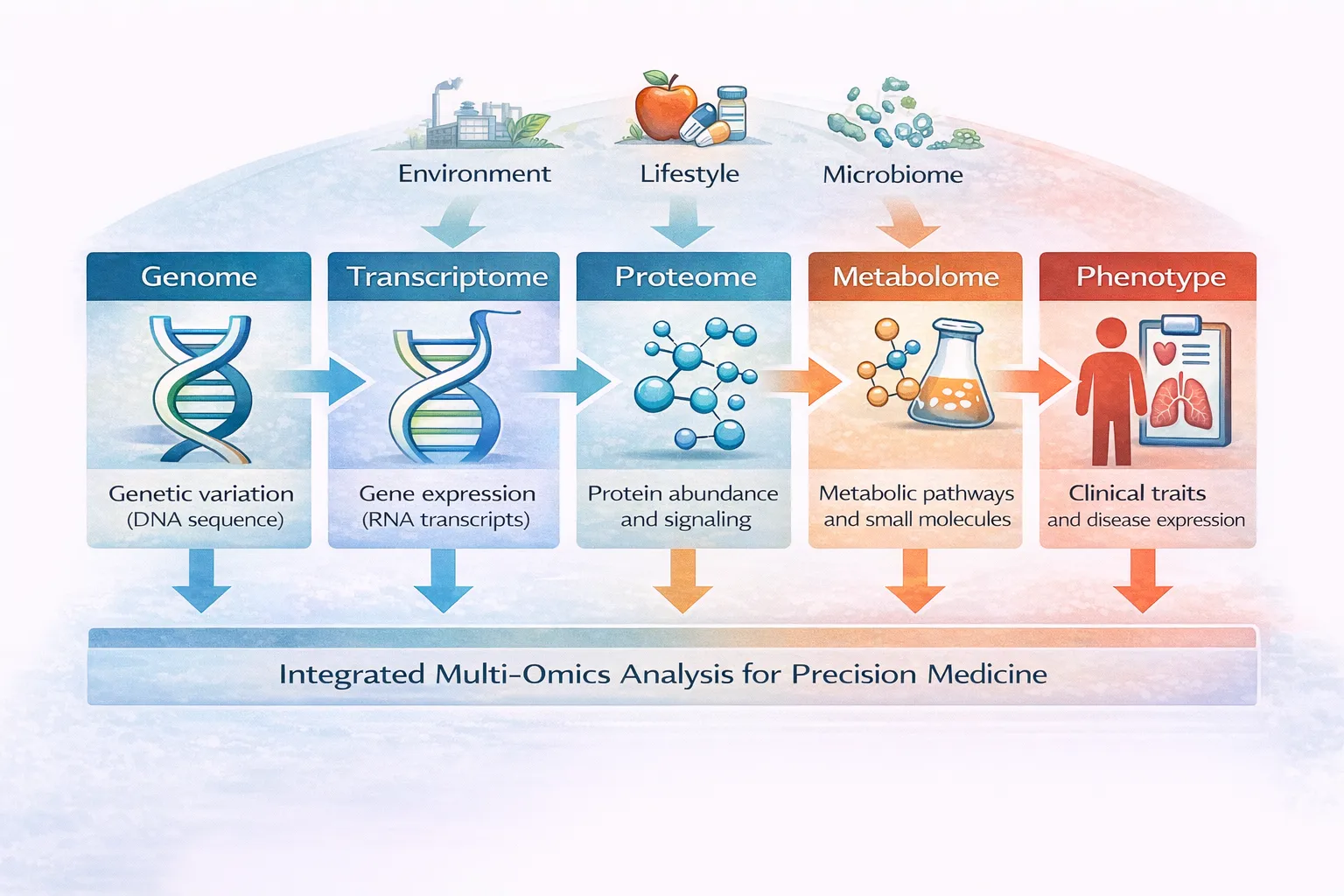
The sequencing of the human genome in the early 2000s provided the first comprehensive map of human genetic information. However, the genome alone cannot fully explain disease. Biological processes are influenced by many layers of information beyond DNA sequence, including:
- Gene expression (transcriptomics)
- Proteins (proteomics)
- Metabolites (metabolomics)
- Epigenetic regulation
- Environmental exposures
Together, these layers form what researchers call multi-omics data. ([Nature][1])
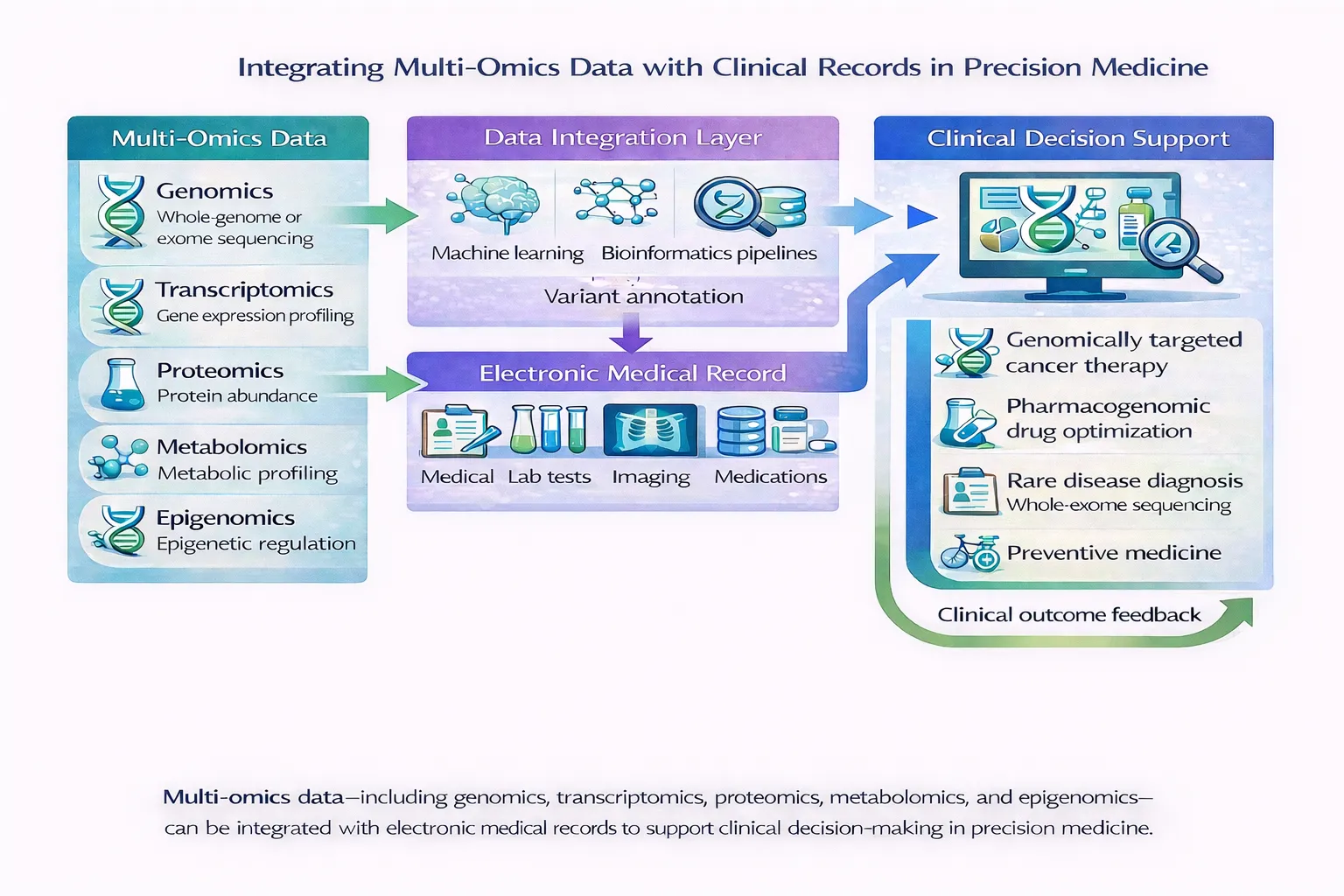
When combined with clinical records and population-scale health data, multi-omics analysis can reveal how biological processes drive disease and how treatments can be tailored to individuals.
The Role of Electronic Health Records in Precision Medicine
Electronic health records are becoming a cornerstone of precision medicine research. They provide longitudinal clinical data such as:
- Diagnoses
- Laboratory results
- Medication history
- Imaging data
- Clinical outcomes
When linked to genomic and molecular data, EHRs allow researchers to connect genetic variation with real-world health outcomes. ([Nature][1])
Example: Linking Genomics to Clinical Outcomes
Large initiatives such as:
- the UK Biobank
- the All of Us Research Program
- the FinnGen project
combine genomic sequencing with detailed medical records from hundreds of thousands to millions of participants. These resources enable researchers to identify genetic factors influencing disease risk, treatment response, and disease progression. ([Nature][1])
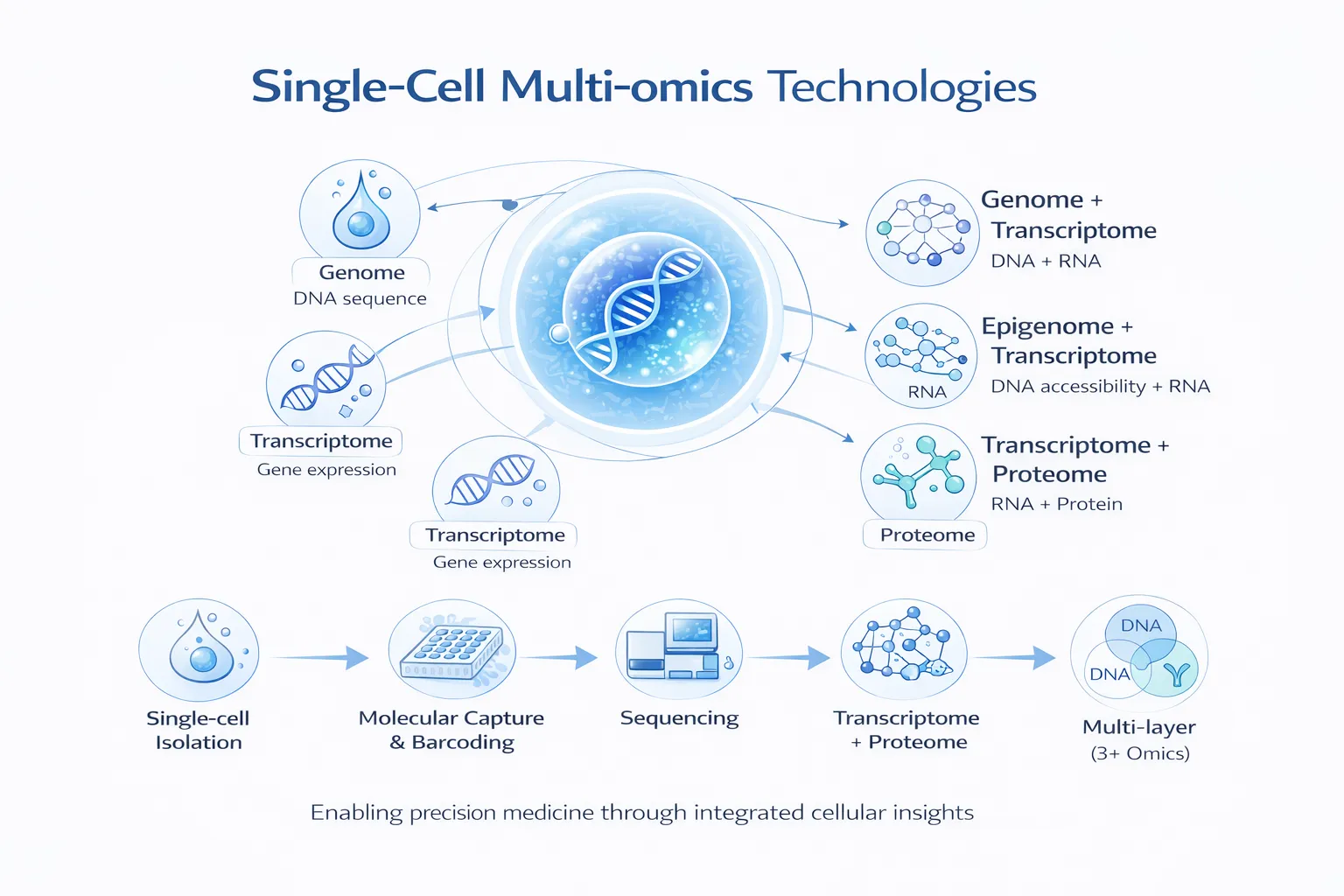
Multi-Omics: Looking Beyond the Genome
While genomics identifies inherited variation, multi-omics data captures dynamic biological processes occurring in cells and tissues.
Examples include:
1. Proteomics
Proteins are the molecules that perform most cellular functions. Changes in protein levels or structure can reveal disease mechanisms or therapeutic targets.
2. Metabolomics
Metabolites are small molecules produced by metabolic reactions. They reflect real-time physiological states and can indicate disease processes earlier than clinical symptoms.
3. Epigenomics
Epigenetic modifications regulate gene activity without altering DNA sequence and can be influenced by lifestyle, environmental exposure, and aging.
Integrating these layers enables a systems-level understanding of disease. ([Nature][1])
Turning Data Into Clinical Insight
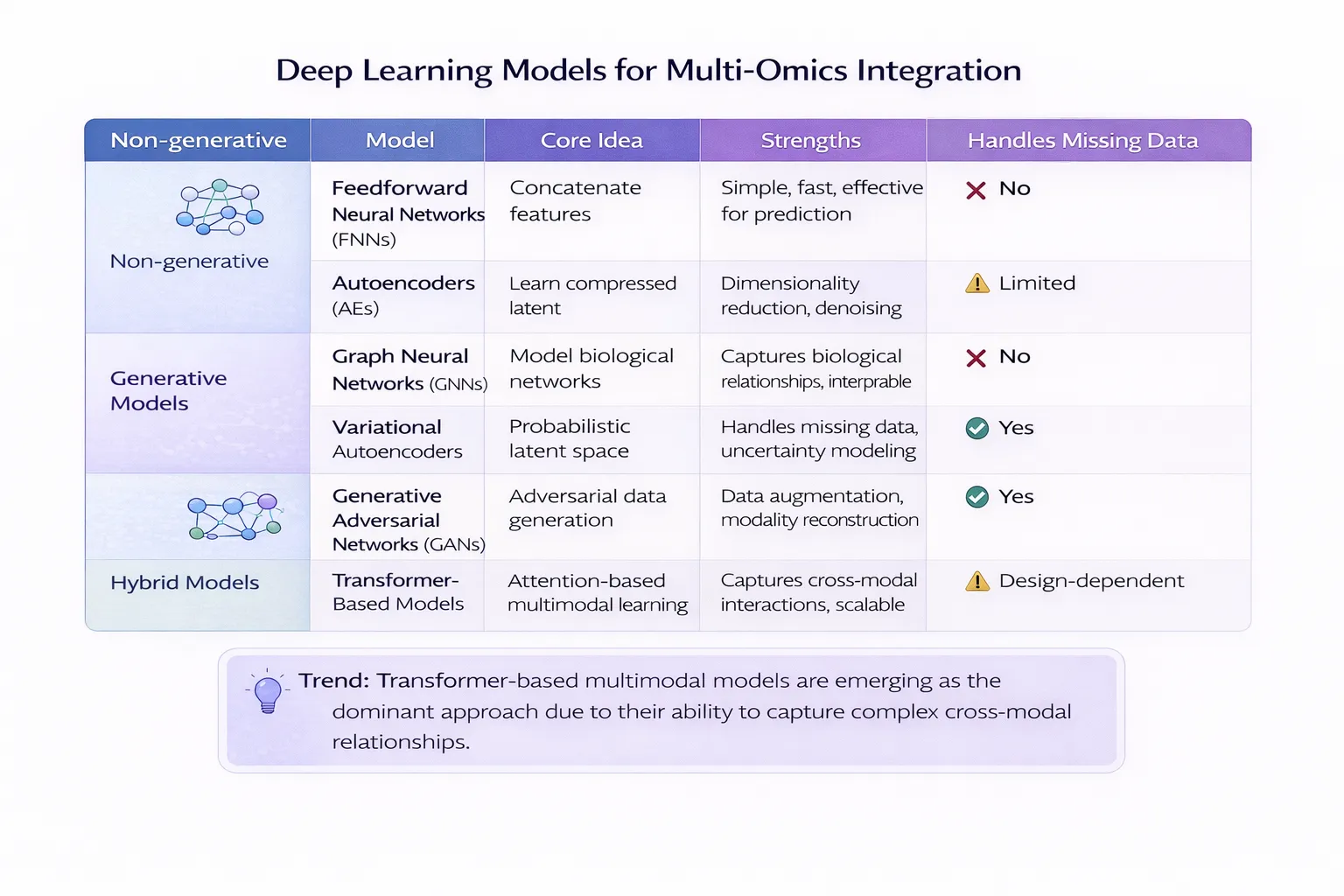
Combining genomic, molecular, and clinical data produces massive datasets that require advanced computational methods to analyze.
Researchers increasingly rely on:
- machine learning
- artificial intelligence
- large-scale statistical modeling
These approaches can uncover patterns linking molecular signatures to disease risk or treatment response.
For example:
- algorithms can identify subtypes of complex diseases such as diabetes
- predictive models can estimate an individual’s risk for cardiovascular disease
- machine learning tools can detect genetic markers associated with drug response
Such analyses help translate biological discoveries into actionable clinical knowledge.
Clinical Applications of Integrated Precision Medicine
Pharmacogenomics
Genetic variation can influence how patients metabolize medications. Pharmacogenomic testing can guide drug selection and dosing.
Example:
- Genetic variants affecting drug metabolism enzymes may change how a patient responds to certain medications.
Early research in pharmacogenetics dates back decades and laid the groundwork for modern personalized drug therapy. ([Nature][1])
Disease Risk Prediction
Large population biobanks allow researchers to identify genetic variants associated with diseases such as:
- Alzheimer’s disease
- cardiovascular disease
- diabetes
Combining these variants into polygenic risk scores may help identify individuals who could benefit from earlier screening or prevention strategies.
Precision Prevention
Integrated health datasets can also identify environmental and lifestyle factors interacting with genetic risk.
For example:
- gene–environment interactions may explain why some individuals develop disease despite similar genetic backgrounds.
Understanding these interactions can inform targeted prevention strategies.
Infrastructure Behind Precision Medicine
Achieving large-scale precision medicine requires extensive research infrastructure.
Key components include:
Biobanks
Population biobanks collect biological samples linked to health data. Examples include:
- UK Biobank
- BioBank Japan
- the Million Veteran Program
These resources enable large-scale genomic research across diverse populations. ([Nature][1])
Data Integration Platforms
Integrating genomic and clinical data requires standardized formats, secure storage, and sophisticated analytics platforms.
Clinical Data Networks
Projects such as the Electronic Medical Records and Genomics (eMERGE) Network link EHR data with genomic sequencing to support both research and clinical implementation. ([Nature][1])
Challenges on the Road to Precision Medicine
Despite rapid progress, several challenges remain.
Data Integration
Different data types—from sequencing data to clinical records—often exist in incompatible formats. Harmonizing these datasets is essential for large-scale analysis.
Population Diversity
Many genomic studies historically focused on populations of European ancestry. Increasing diversity in genomic research is critical for equitable precision medicine. ([Nature][1])
Ethical and Privacy Considerations
Large health datasets raise important questions about:
- data security
- patient consent
- responsible data sharing
Addressing these issues is essential to maintaining public trust.
The Future of Precision Medicine
Precision medicine is moving toward a continuous learning healthcare system, where clinical data, genomic information, and research insights feed back into patient care.
In this model:
- Patients contribute clinical and molecular data.
- Researchers analyze patterns across populations.
- Insights are translated into clinical guidelines.
- Clinicians apply these insights to improve patient care.
As genomic technologies, multi-omics profiling, and AI analytics continue to advance, healthcare will increasingly move toward predictive, preventive, and personalized care.