Introduction
Advances in genomic science have made it possible not only to read DNA but also to modify it with remarkable precision. This capability—known as gene editing—allows scientists to alter genetic sequences within living cells. In medicine, gene editing offers the potential to treat diseases at their genetic root cause, rather than managing symptoms alone.
Over the past two decades, several gene editing technologies have emerged, each with different strengths and limitations. These tools are now being used in research laboratories, clinical trials, and in some cases early therapeutic applications.
This article introduces the major gene editing technologies, explains how they work, and explores their growing role in precision medicine.
What Is Gene Editing?
Gene editing refers to technologies that allow researchers to modify DNA sequences at specific locations in the genome. These modifications can include:
- Deleting a gene
- Correcting a mutation
- Inserting new genetic material
- Regulating gene expression
Gene editing works by creating targeted changes to DNA. Most systems operate by:
- Identifying a specific DNA sequence
- Cutting or modifying the DNA at that location
- Allowing the cell’s natural repair mechanisms to complete the edit
These capabilities make gene editing a powerful approach for studying gene function and developing therapies for genetic diseases.
Below is an updated and expanded draft article incorporating the structured comparison framework (mechanism, timeline, matrix comparison, and clinical examples) that aligns with the gene editing technologies comparison figure you generated.
Gene Editing Technologies: From ZFNs to CRISPR and Beyond
Gene editing technologies allow scientists to modify DNA within living cells, enabling the correction of disease-causing mutations, the study of gene function, and the development of new therapies. Over the past three decades, genome engineering has evolved from complex protein-based tools to highly precise RNA-guided systems.
Today, technologies such as CRISPR, base editing, and prime editing are transforming biomedical research and clinical medicine, forming a cornerstone of precision medicine.
This article introduces the major gene editing platforms used in research and medicine and compares their mechanisms, capabilities, and clinical applications.
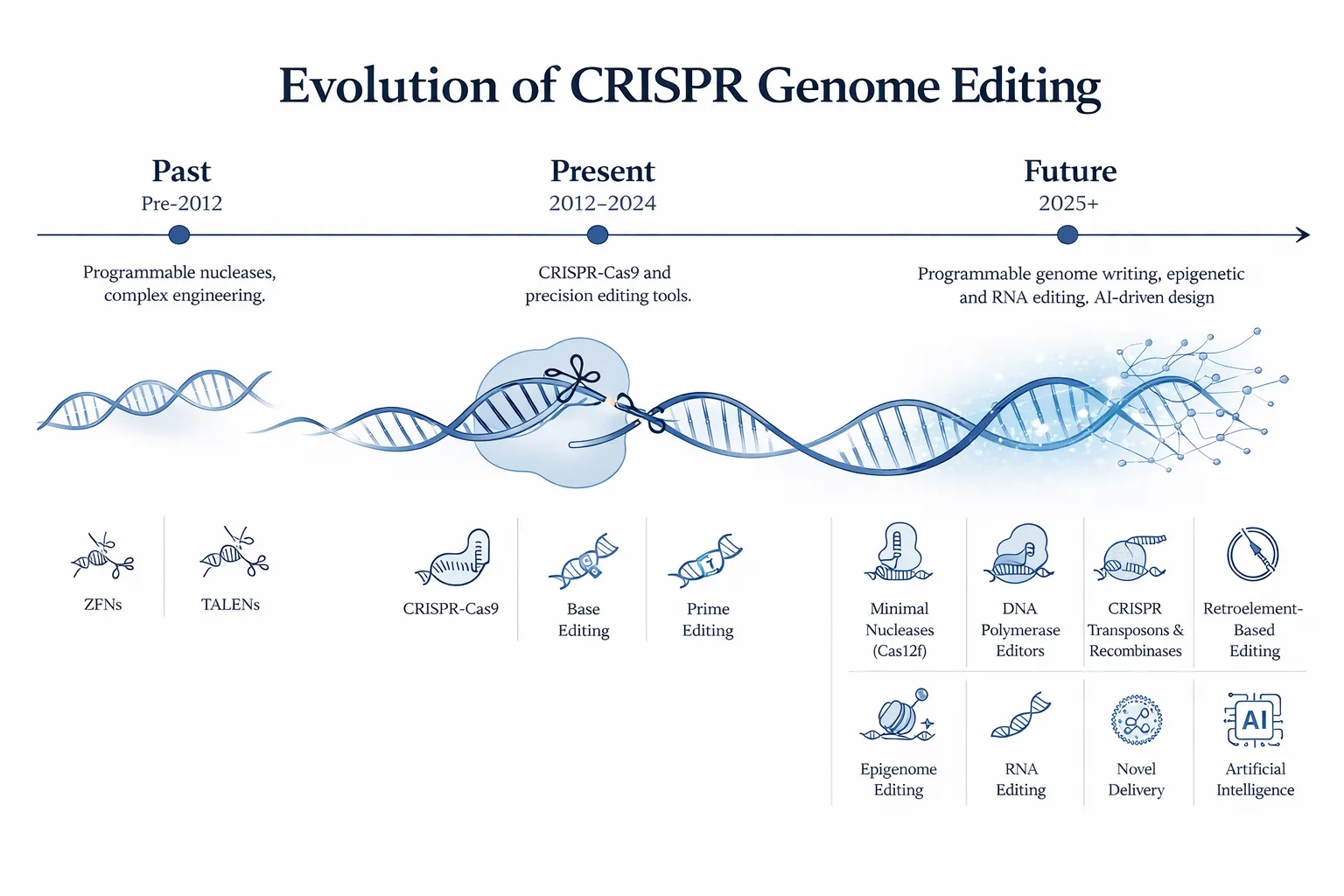
The Evolution of Gene Editing Technologies
Modern gene editing has progressed through several key technological milestones:
| Era | Technology | Key Innovation |
|---|---|---|
| 1990s | Zinc Finger Nucleases (ZFN) | First programmable DNA-targeting nucleases |
| 2000s | TALENs | Modular protein-DNA targeting system |
| 2012 | CRISPR-Cas9 | RNA-guided genome editing |
| 2016 | Base Editing | Precise single base conversion without DNA cutting |
| 2019 | Prime Editing | Flexible editing capable of insertions, deletions, and substitutions |
Each generation of tools has improved the precision, flexibility, and ease of genome editing.
How Gene Editing Works
Most gene editing technologies operate using a two-step process:
- Target recognition – identifying the DNA sequence to be edited.
- DNA modification – cutting or chemically modifying the DNA to introduce changes.
Earlier tools relied on engineered proteins to recognize DNA sequences, while newer methods use RNA molecules to guide the editing machinery.
Zinc Finger Nucleases (ZFN)
Zinc finger nucleases were among the first programmable gene editing tools developed in the 1990s.
They combine:
- Zinc finger DNA-binding proteins
- FokI nuclease enzymes that cut DNA
When two zinc finger nucleases bind adjacent DNA sequences, the nuclease domains create a double-strand break, triggering the cell’s DNA repair mechanisms.
Advantages
- Highly specific targeting when properly engineered
Limitations
- Difficult and time-consuming to design
- Limited scalability
Clinical Example
Early gene therapy trials used ZFN technology to edit immune cells in HIV patients, attempting to disrupt the CCR5 gene, which the virus uses to infect cells.
TALENs (Transcription Activator-Like Effector Nucleases)
TALENs were developed in the 2000s as a more flexible alternative to ZFNs.
They use:
- TALE DNA-binding domains
- FokI nuclease cleavage domains
TALENs can be engineered to recognize almost any DNA sequence, making them easier to design than zinc finger systems.
Advantages
- High targeting specificity
- Easier customization than ZFNs
Limitations
- Large protein size makes delivery difficult
- Slower engineering compared with CRISPR
Clinical Example
TALEN technology has been used to engineer CAR-T immune cells for cancer therapy, improving immune cell targeting of tumours.
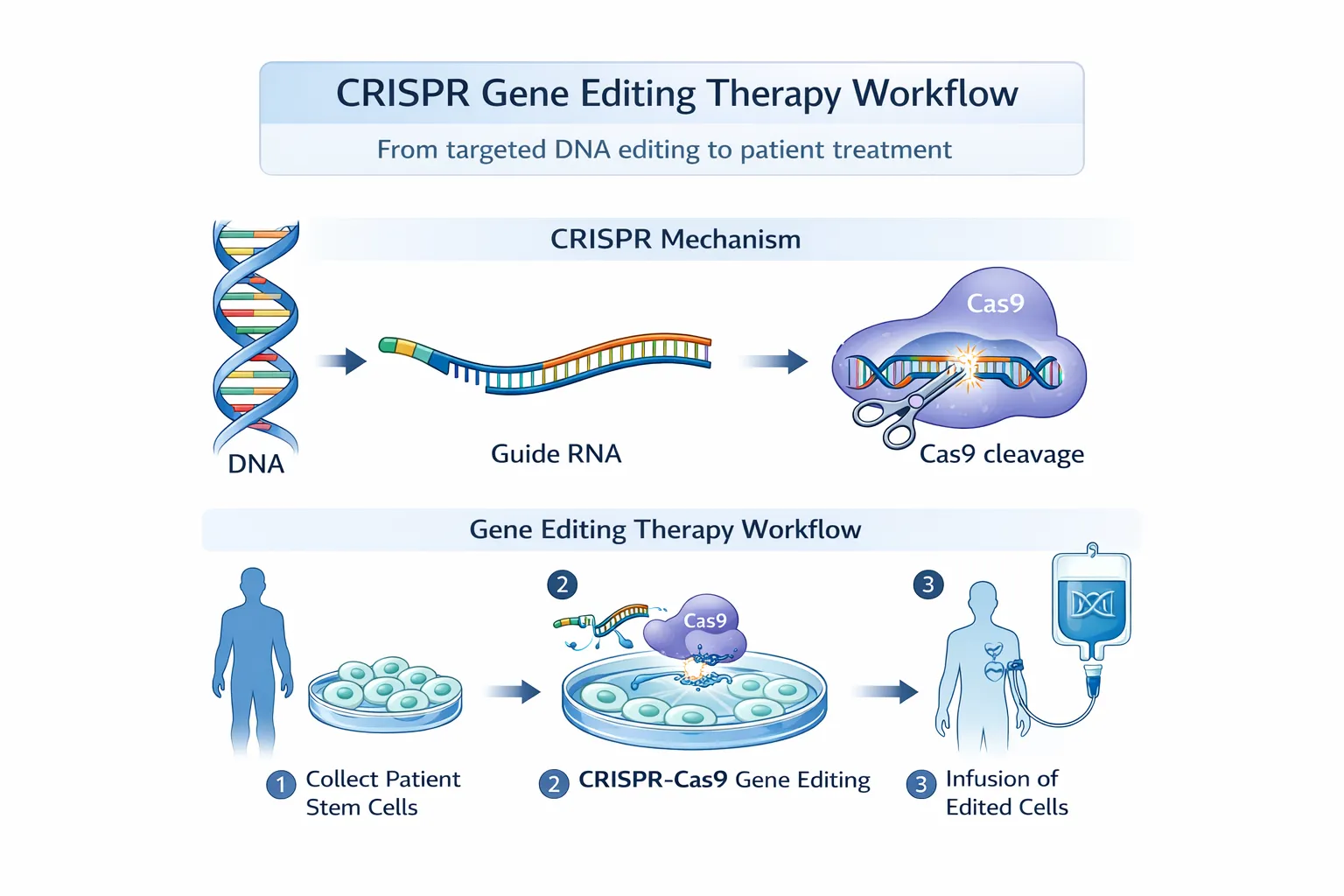
CRISPR-Cas9
CRISPR technology revolutionized gene editing when it was introduced in 2012.
Unlike earlier technologies, CRISPR uses:
- a guide RNA (gRNA) to locate the DNA target
- the Cas9 enzyme to cut DNA
This system is much easier to program than protein-based editors.
Advantages
- Simple design
- Highly scalable
- Relatively inexpensive
Limitations
- Can produce unintended off-target edits
- Double-strand DNA breaks may introduce unwanted mutations
Clinical Example
CRISPR-based therapies are being developed to treat sickle cell disease, where edited stem cells are reinfused into patients to restore healthy red blood cell production.
Base Editing
Base editing is a next-generation CRISPR-derived technology that enables precise single-nucleotide changes without cutting DNA.
Instead of breaking the DNA strand, base editors chemically convert one base to another.
Examples include:
- Cytosine base editors (C → T)
- Adenine base editors (A → G)
Advantages
- High precision
- Reduced risk of DNA damage
- Efficient for correcting point mutations
Limitations
- Only certain base conversions are possible
- Off-target RNA editing can occur
Clinical Example
Base editing is being investigated for genetic liver diseases and metabolic disorders caused by single base mutations.
Prime Editing
Prime editing is one of the most flexible gene editing technologies developed to date.
It combines:
- a modified Cas9 enzyme
- a reverse transcriptase enzyme
- a prime editing guide RNA (pegRNA)
This system can perform:
- base substitutions
- insertions
- deletions
without creating double-strand DNA breaks.
Advantages
- Very precise editing
- Broad range of possible DNA changes
- Lower risk of unintended mutations
Limitations
- Technically complex
- Still under active research
Clinical Potential
Prime editing may eventually allow correction of many disease-causing genetic mutations, including those responsible for rare inherited disorders.
Comparison of Gene Editing Technologies
The major editing platforms differ in their mechanism, precision, and clinical maturity, as well as the types of clinical applications where they are most useful.
| Feature | ZFN | TALEN | CRISPR | Base Editing | Prime Editing |
|---|---|---|---|---|---|
| Targeting method | Protein-DNA recognition | Protein-DNA recognition | RNA-guided targeting | RNA-guided deaminase | RNA-guided reverse transcriptase |
| Double-strand DNA break | Yes | Yes | Yes | No | No |
| Editing precision | Moderate | High | High | Very high | Very high |
| Ease of design | Difficult | Moderate | Easy | Moderate | Moderate |
| Typical mutation | Insertions/deletions | Insertions/deletions | Insertions/deletions | Single base change | Insert/delete/substitute |
| Clinical scenarios | Early gene therapy research; HIV resistance via CCR5 disruption | Engineered immune cells (CAR-T therapy), cancer immunotherapy | Sickle cell disease, beta-thalassemia, cancer cell engineering | Correction of single-nucleotide mutations (e.g., metabolic or liver diseases) | Potential correction of complex genetic mutations in rare inherited diseases |
Explanation of Clinical Applications
ZFN One of the earliest tools used in human trials. For example, ZFN editing of the CCR5 gene in T cells has been explored to make immune cells resistant to HIV infection.
TALEN Commonly used for engineering immune cells, particularly in the development of CAR-T therapies for cancer treatment.
CRISPR Currently the most widely used clinical gene editing platform, with approved or late-stage therapies for conditions such as sickle cell disease and beta-thalassemia.
Base Editing Particularly suited to diseases caused by single nucleotide mutations, which account for a large proportion of inherited genetic disorders.
Prime Editing Still largely experimental but potentially capable of correcting many types of mutations, including insertions, deletions, and substitutions associated with rare genetic diseases.
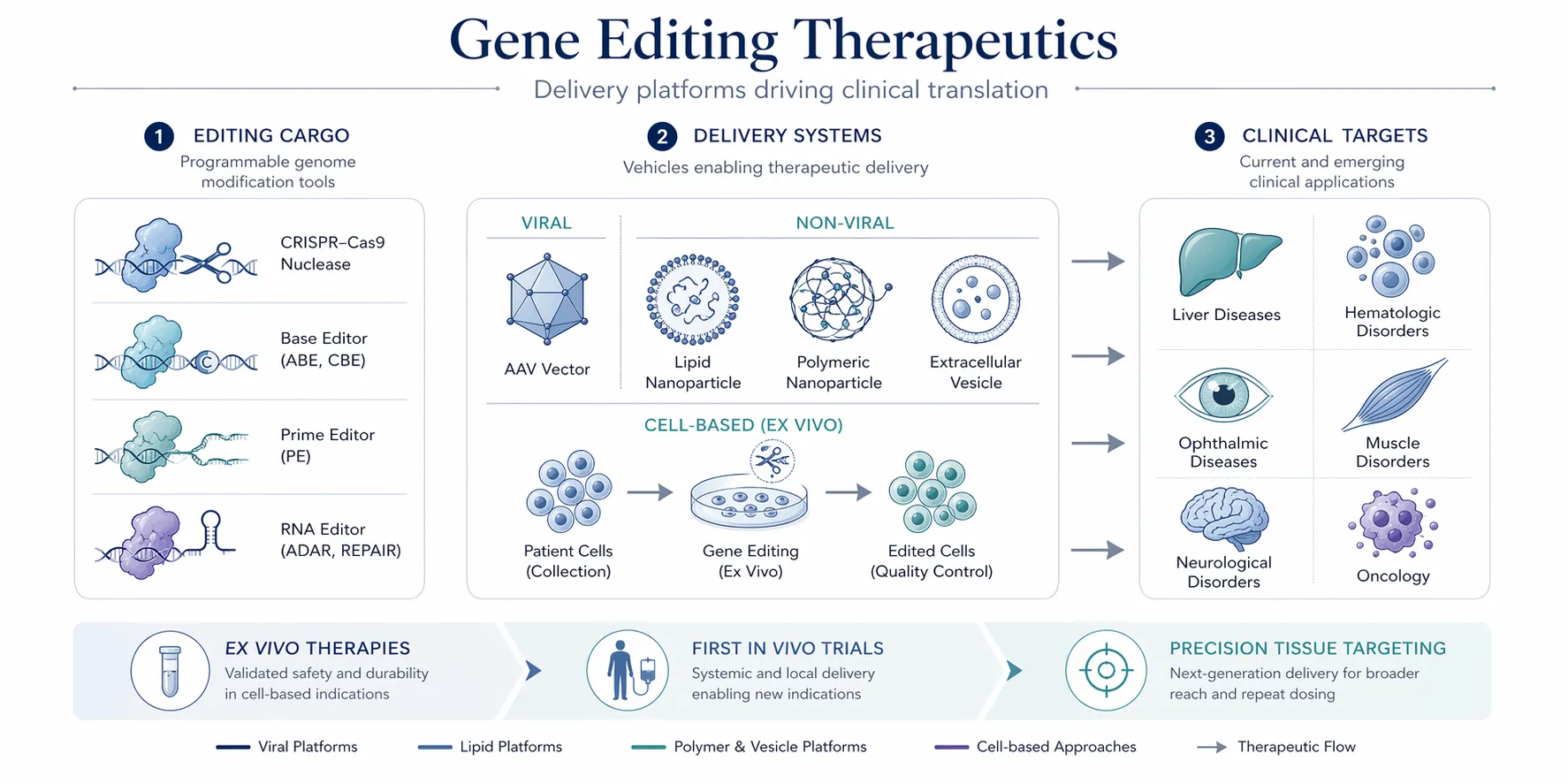
Gene Editing in Medicine
Gene editing technologies are already being applied in several medical areas:
Genetic Disease Treatment
Editing defective genes may allow curative therapies for conditions such as:
- sickle cell disease
- beta-thalassemia
- inherited blindness
Cancer Therapy
Engineered immune cells can be edited to improve tumour recognition, forming the basis of next-generation CAR-T therapies.
Infectious Disease Research
Gene editing can modify immune cells or viral receptors to study viral infection mechanisms.
Challenges and Ethical Considerations
Despite rapid progress, gene editing raises important scientific and ethical questions.
Safety
Potential risks include:
- unintended off-target mutations
- immune responses
- incomplete editing
Germline Editing
Editing embryos or reproductive cells raises ethical concerns because changes could be inherited by future generations.
Accessibility
Advanced gene therapies may initially be expensive and limited to specialised centres, raising questions about equitable access.
The Future of Gene Editing
Gene editing technologies continue to evolve rapidly.
Emerging areas of research include:
- improved precision editing systems
- epigenome editing
- RNA editing
- in vivo gene editing therapies
As these technologies mature, they are likely to play an increasingly important role in precision medicine, enabling treatments tailored to the genetic causes of disease.