Introduction
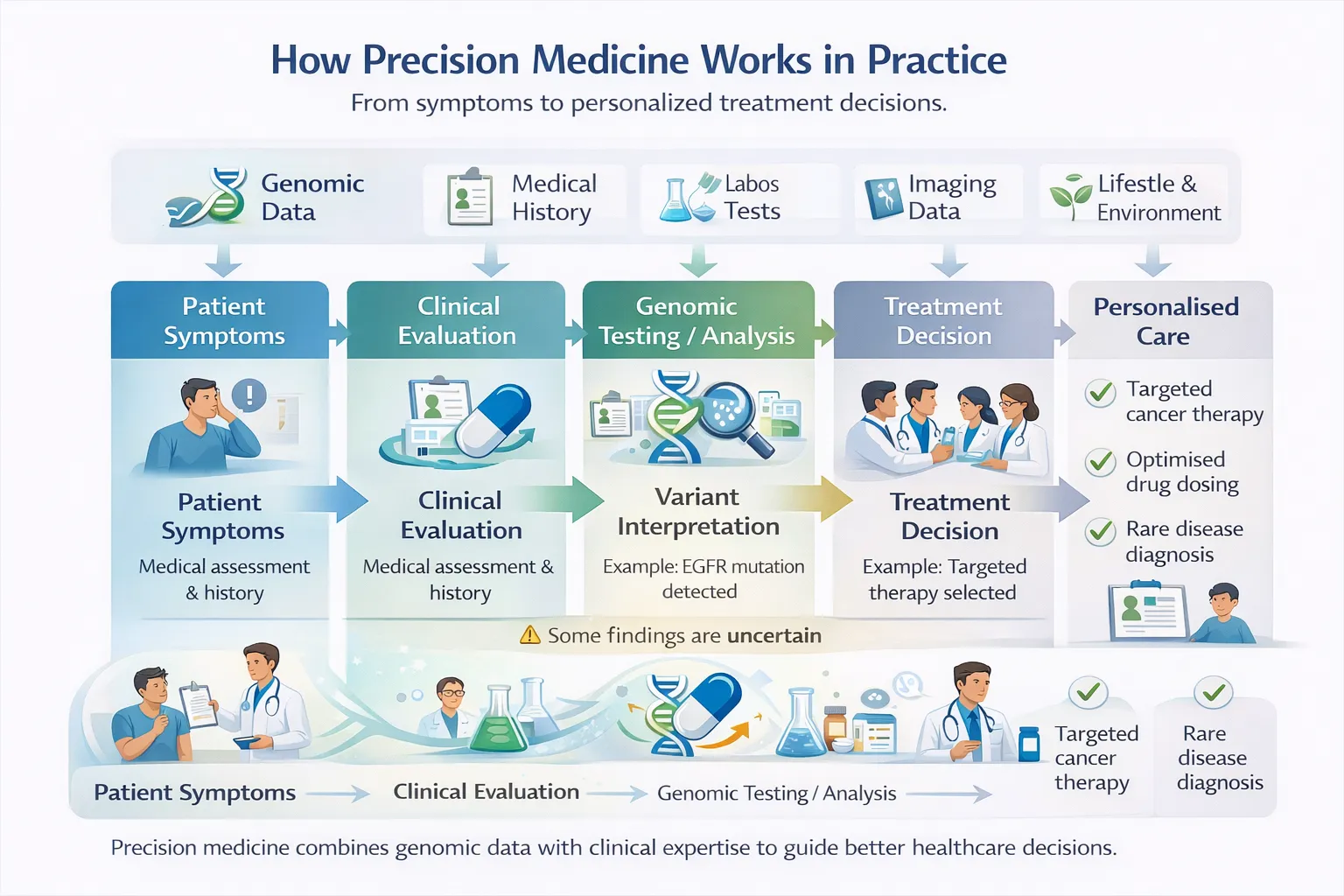
Precision medicine is often described as “the right treatment for the right patient at the right time.” But what actually happens between a patient’s DNA sample and a clinical decision?
This section walks through the real-world clinical workflow—step by step.
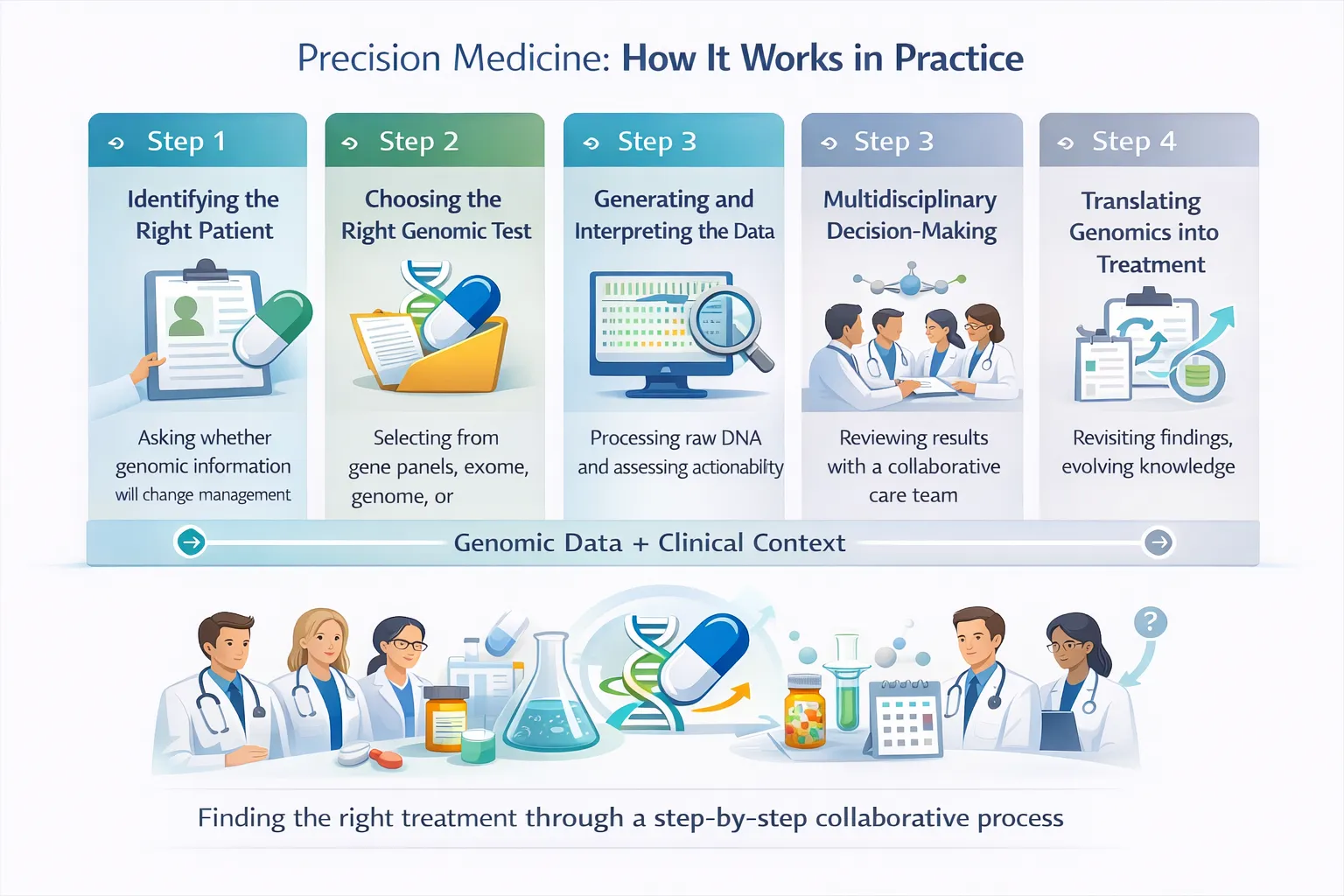
Step 1: Identifying the Right Patient
Precision medicine does not start with sequencing everyone. It starts with asking the right clinical question.
Common entry points include:
- A patient who has failed standard therapy
- A cancer that is advanced, recurrent, or atypical
- A child or adult with a suspected rare genetic disorder
- A patient starting a medication known to have genetic dosing or safety considerations
For patients, this often feels like:
“My doctor says there’s a genetic test that might help.”
For clinicians, it begins with:
“Will genomic information meaningfully change management?”
Step 2: Choosing the Right Genomic Test
Not all genomic tests are the same. Selecting the wrong test can produce confusing or unusable results.
Common clinical options include:
Targeted gene panels Focused testing for specific genes (e.g. cancer driver mutations or pharmacogenes)
Whole exome sequencing (WES) Looks at all protein-coding genes (~1–2% of the genome)
Whole genome sequencing (WGS) Captures coding and non-coding regions, structural variants, and regulatory elements
Pharmacogenomic (PGx) panels Designed specifically to guide drug choice and dosing
Key clinical principle:
More data is not always better—appropriate data is.
Step 3: Generating and Interpreting the Data
Once sequencing is completed, raw DNA data is processed through bioinformatic pipelines to identify genetic variants.
This is where expectations often diverge:
What patients often expect
- A clear answer
- A definitive diagnosis
- A single “actionable gene”
What clinicians usually see
- Dozens to thousands of variants
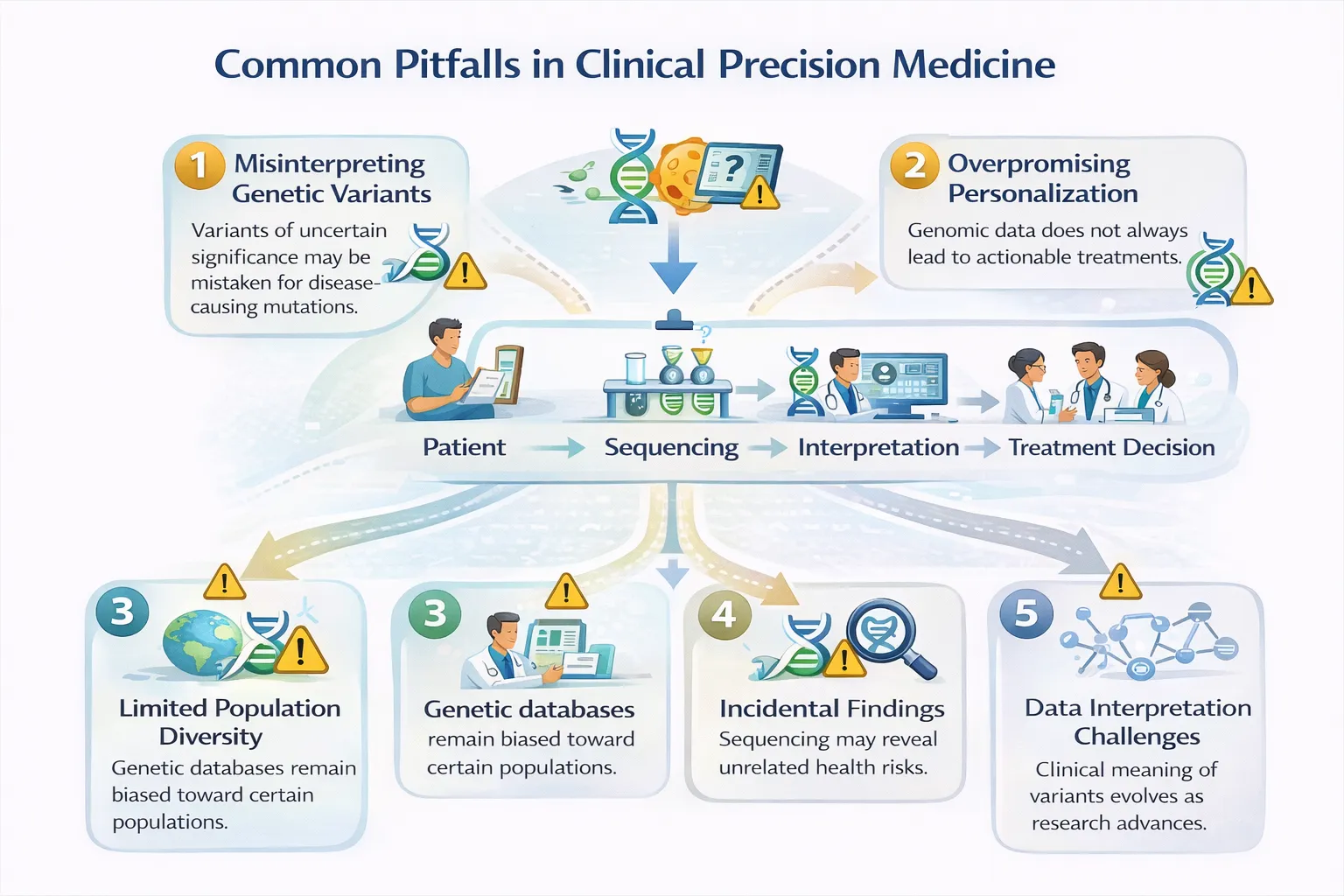
- Many classified as variants of uncertain significance (VUS)
- Actionability that depends on evidence strength, context, and guidelines
Interpretation typically relies on:
- Population databases
- Functional studies
- Clinical correlation
- Expert consensus guidelines such as those from Clinical Pharmacogenetics Implementation Consortium or national genomic programs
Step 4: Multidisciplinary Decision-Making
Precision medicine is rarely a solo act.
Clinical decisions often involve:
- Treating physicians
- Clinical geneticists
- Molecular pathologists
- Pharmacists
- Genetic counsellors
- Molecular tumour boards (in oncology)
Together, the team asks:
- Is this variant clinically actionable?
- Does it explain the patient’s presentation?
- Is there a validated treatment, dose adjustment, or trial option?
- Do benefits outweigh risks and uncertainty?
This collaborative step is one of the most underappreciated aspects of precision medicine.
Step 5: Translating Genomics into Treatment
When genomic information is actionable, it may lead to:
Pharmacogenomic decisions
- Dose adjustment
- Drug avoidance
- Alternative therapy selection
Example:
- CYP2C19 variants influencing clopidogrel effectiveness
- HLA variants predicting severe drug hypersensitivity
Guidance may align with recommendations recognized by regulators such as the U.S. Food and Drug Administration or international expert panels.
Cancer treatment selection
- Targeted therapies based on tumour mutations
- Avoidance of ineffective or toxic treatments
- Eligibility for genotype-matched clinical trials
Importantly, the tumour genome, not the patient’s inherited DNA, often drives these decisions.
Rare disease diagnosis
- Ending diagnostic odysseys
- Informing prognosis and family planning
- Occasionally enabling gene-targeted or repurposed therapies
Even when no treatment exists, a diagnosis can still be transformative.
Step 6: Follow-Up, Re-analysis, and Limits
Precision medicine is not a one-time event.
- Genomic interpretations change as science advances
- Variants may be reclassified
- New therapies may emerge
- Not all results lead to action
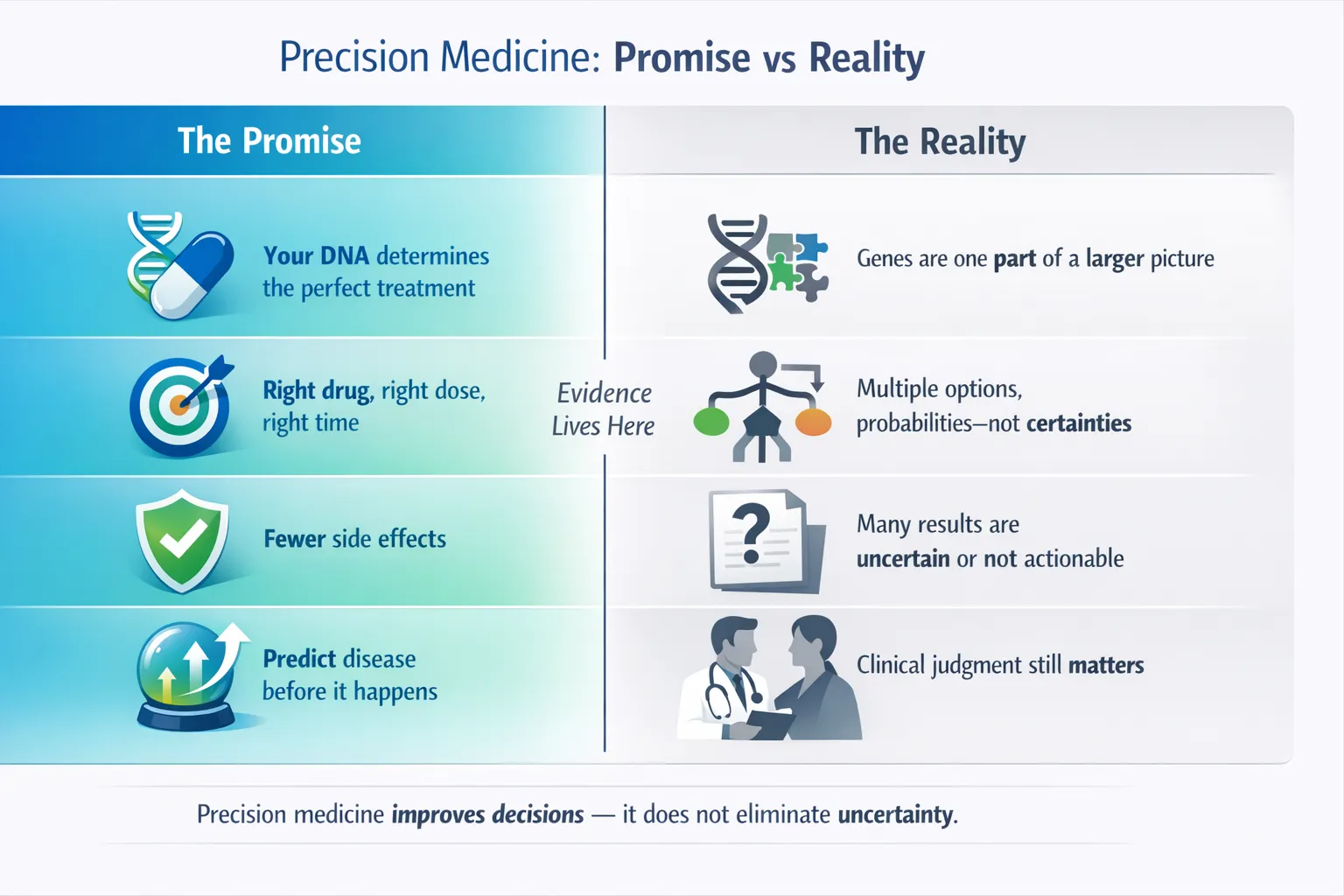
Crucial reality check: Precision medicine increases clarity and probability, not certainty.
What This Means in Real Life
For patients:
- Fewer trial-and-error treatments
- More informed decisions
- Better understanding of risk and benefit
- Occasionally, life-changing answers
For clinicians:
- More data—but also more responsibility
- A need for genomic literacy and decision support
- Recognition that precision medicine complements, rather than replaces, clinical judgment
A Final Perspective
Precision medicine works best when:
- The clinical question is clear
- The test is appropriate
- Interpretation is cautious
- Decisions are collaborative
- Expectations are realistic
In practice, precision medicine is less about perfect prediction and more about making better-informed choices in complex situations.