How Integrating Biological Data Is Transforming Healthcare
Modern medicine is undergoing a profound transformation. Instead of treating diseases based only on symptoms or population averages, clinicians are increasingly able to tailor healthcare to the biological profile of each individual patient. This approach—known as precision medicine—draws on advances in genomics, bioinformatics, and multi-omics technologies to better understand health and disease. ([nature.com][1])
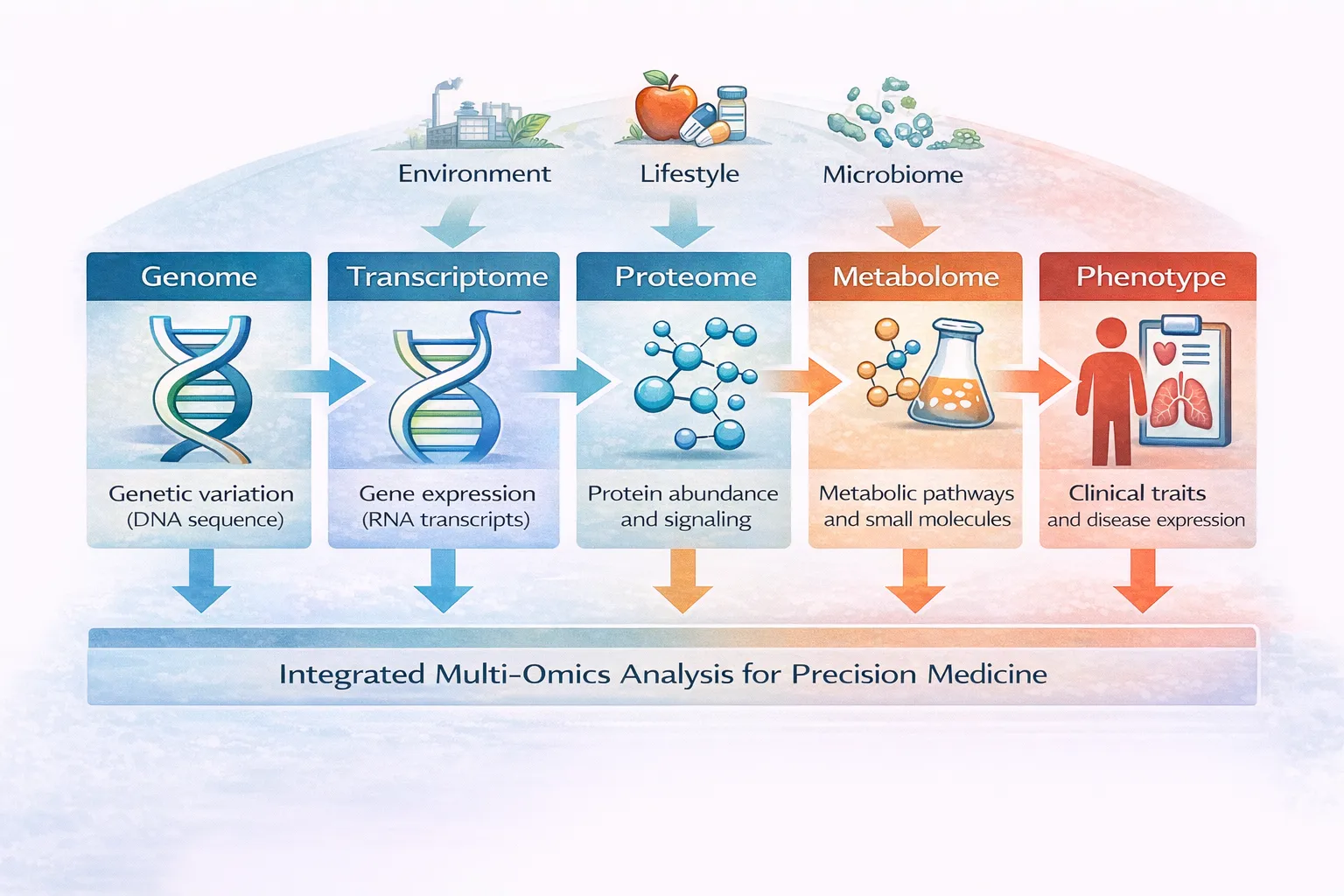
In recent years, researchers have realized that studying DNA alone is not enough. Human health arises from complex interactions among genes, proteins, metabolism, environmental exposures, and lifestyle factors. Integrating these different layers of biological information—collectively called multi-omics—is now one of the most powerful approaches in biomedical science.
This article explains how genomics and multi-omics are shaping the future of precision medicine, particularly in pediatric healthcare.
From the Human Genome to Precision Medicine
The modern era of genomics began with the Human Genome Project, completed in 2003. The project produced the first reference sequence of the human genome and revealed that humans have roughly 20,000–25,000 protein-coding genes. ([Nature][1])
This achievement revolutionized biomedical research. Scientists gained a new understanding of the genetic basis of disease and began exploring how genetic variation influences health, treatment response, and disease risk.
However, it soon became clear that genes alone do not determine disease. Environmental exposures, diet, lifestyle, and biological regulation all influence how genes function in the body. Precision medicine therefore seeks to integrate multiple biological layers to create a complete picture of human health.
Precision medicine aims to:
- Predict disease risk
- Detect disease earlier
- Select the most effective treatments
- Minimize adverse drug reactions
- Improve long-term health outcomes
This represents a shift from reactive medicine to proactive, preventive healthcare. ([Nature][1])
The Rise of Multi-Omics
“Omics” refers to technologies that study large sets of biological molecules in cells or tissues. Modern precision medicine combines several of these data layers.
Key Omics Technologies
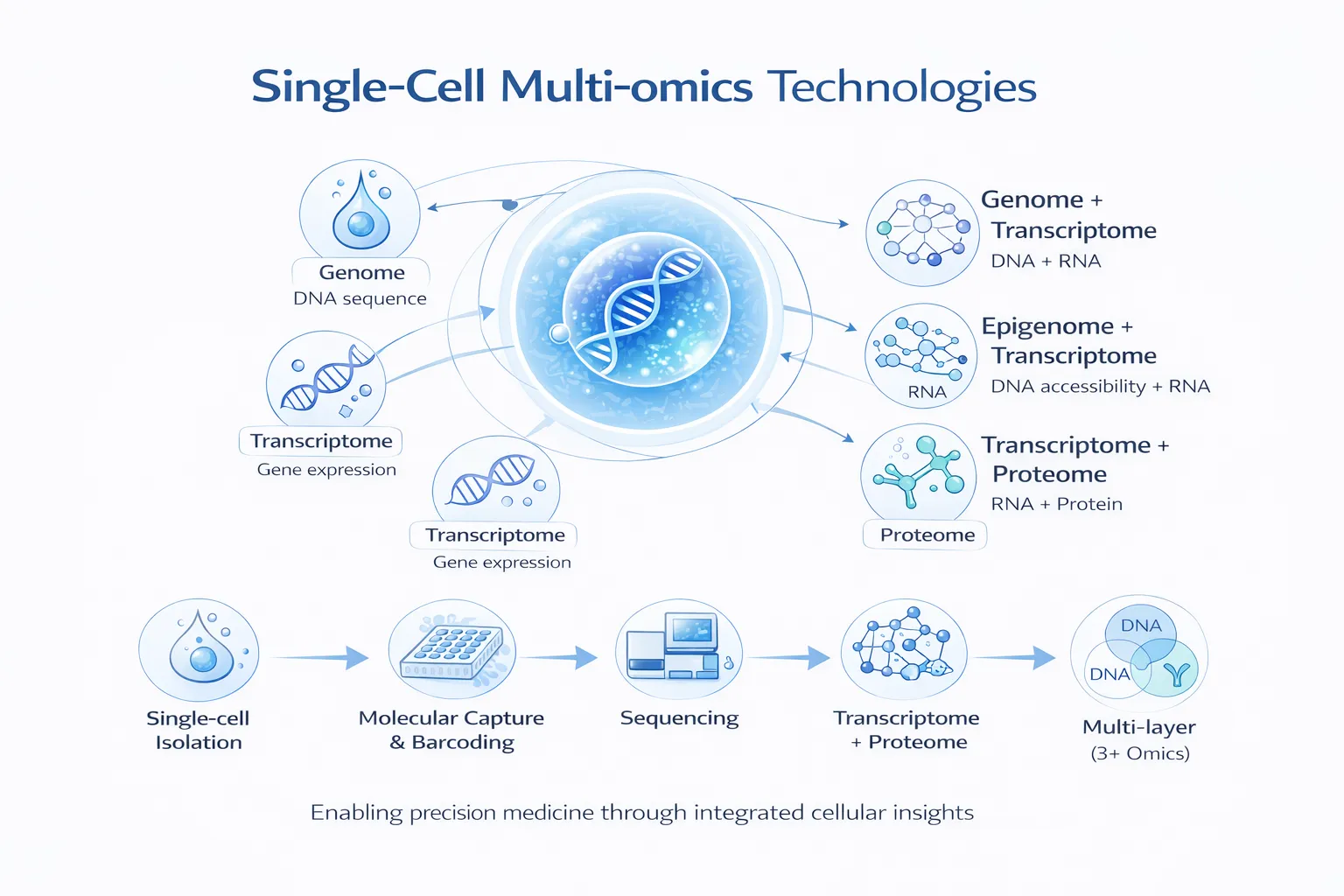
Genomics The study of DNA sequences and genetic variation. Advances in next-generation sequencing (NGS) have made genome sequencing faster and far less expensive than in the past. ([Nature][1])
Transcriptomics Examines RNA molecules to determine which genes are actively expressed in cells.
Proteomics Studies proteins—the molecules responsible for most biological functions.
Metabolomics Measures small molecules produced during metabolism, offering insight into real-time cellular activity.
Epigenomics Investigates regulatory modifications to DNA (such as DNA methylation) that affect gene activity without changing the genetic code.
Exposomics Examines environmental exposures such as diet, chemicals, pollution, and lifestyle factors that influence health.
Each of these fields provides a different view of biology. When integrated, they can reveal disease mechanisms that would otherwise remain hidden.
Why Integrating Multi-Omics Matters
Analyzing a single type of biological data often provides only limited insight. Multi-omics integration allows researchers to connect genetic variation with downstream biological effects, such as changes in gene expression or metabolic pathways. ([Nature][1])
For example:
- Genetic variants may alter RNA expression patterns
- RNA changes may affect protein levels
- Protein changes can influence metabolic pathways
- Environmental exposures may modify gene regulation through epigenetic mechanisms
By combining these datasets, scientists can identify disease subtypes, biomarkers, and therapeutic targets that would otherwise be difficult to detect.
Advances in Sequencing Technologies
One major driver of precision medicine has been the rapid evolution of sequencing technologies.
Next-Generation Sequencing (NGS)
NGS platforms can sequence millions of DNA fragments simultaneously, enabling:
- Whole-genome sequencing
- Whole-exome sequencing
- RNA sequencing
These technologies have dramatically reduced the cost of sequencing—from thousands of dollars per genome to only a few hundred dollars in recent years. ([Nature][1])
Long-Read Sequencing
Newer sequencing methods such as:
- Single-molecule real-time sequencing (SMRT)
- Nanopore sequencing
allow scientists to read much longer DNA fragments and detect complex structural variants or epigenetic modifications. ([Nature][1])
These technologies are increasingly important for diagnosing genetic diseases that were previously difficult to detect.
Real-World Clinical Applications
Multi-omics research is already transforming clinical practice in several areas.
Pediatric Oncology
Cancer is fundamentally a genetic disease. Multi-omics approaches can identify:
- oncogenic mutations
- gene fusions
- transcriptional signatures
These insights help clinicians select targeted therapies tailored to the tumor’s molecular profile.
For example, RNA sequencing has been shown to identify gene fusions and mutations in pediatric leukemia that were missed by traditional diagnostic methods. ([Nature][1])
Rare Disease Diagnosis
Many rare genetic disorders remain undiagnosed using conventional testing.
Combining genome sequencing with transcriptomics and proteomics can:
- reveal pathogenic variants
- clarify variant significance
- improve diagnostic yield
Studies suggest RNA sequencing may resolve 7–35% of previously undiagnosed genetic conditions. ([Nature][1])
Metabolic Disorders
Metabolomics can detect abnormal metabolic pathways and identify biomarkers for diseases such as autism spectrum disorders or inherited metabolic conditions.
One study found that untargeted metabolomic screening identified inborn metabolic disorders at a rate six times higher than traditional screening methods. ([Nature][1])
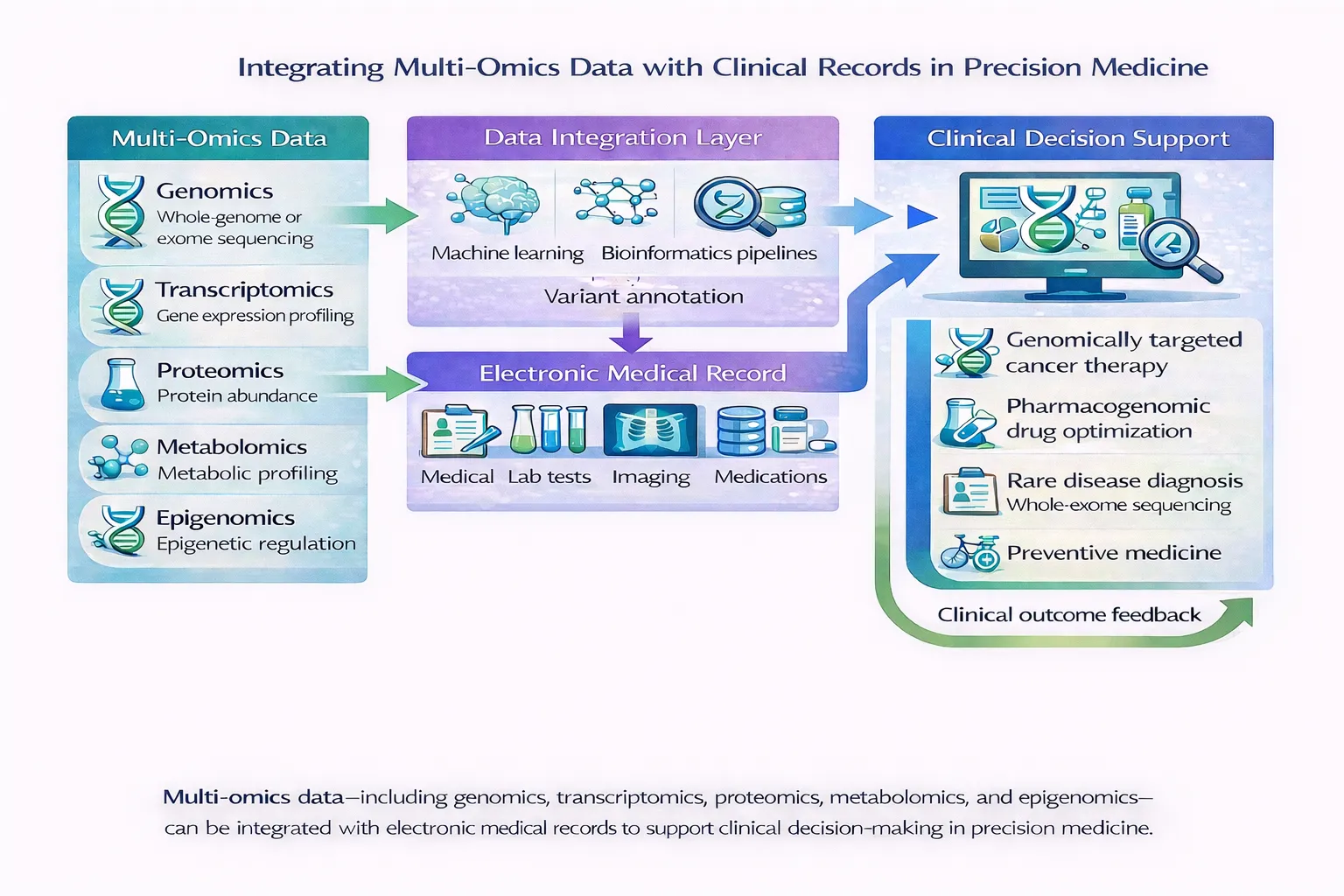
Integrating Multi-Omics with Electronic Medical Records
Precision medicine becomes most powerful when biological data are combined with clinical information.
Electronic health records (EHRs) contain both:
- structured data (lab tests, medications, vital signs)
- unstructured data (clinical notes, imaging reports)
Integrating EHR data with multi-omics allows clinicians to interpret genomic findings in the context of the patient’s medical history and clinical presentation. ([Nature][1])
Emerging technologies such as FHIR standards, artificial intelligence, and machine learning are helping automate this integration and enable real-time clinical decision support.
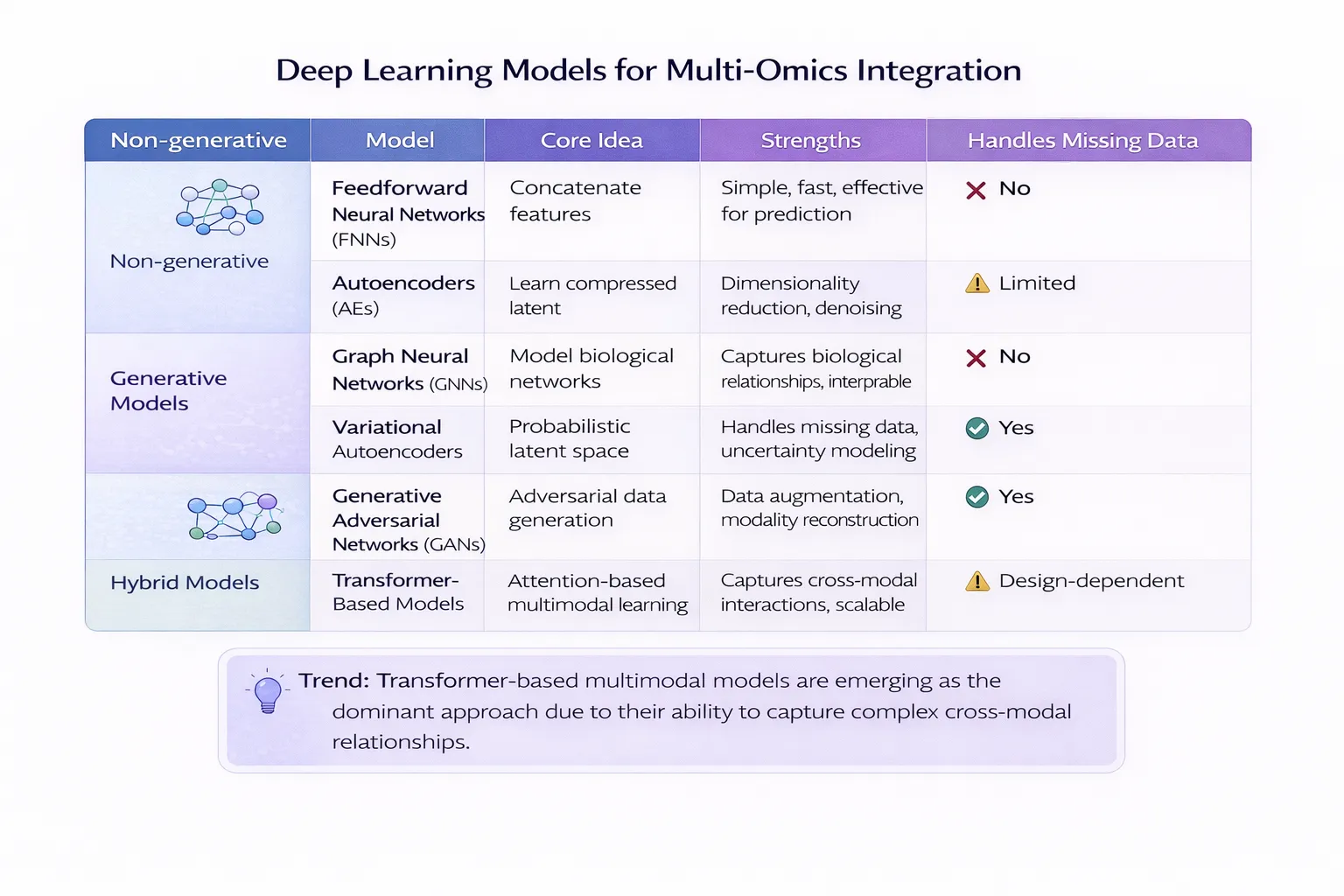
The Role of Bioinformatics and Artificial Intelligence
Multi-omics datasets are extremely large and complex. Bioinformatics tools and AI algorithms are essential for extracting clinically meaningful information.
These tools help researchers:
- identify disease biomarkers
- classify disease subtypes
- discover new therapeutic targets
- predict treatment responses
Large genomic databases—such as gnomAD, ClinVar, OMIM, and ClinGen—are also crucial for interpreting genetic variants in clinical testing. ([Nature][1])
Challenges and Ethical Considerations
Despite its promise, precision medicine still faces several challenges.
Data Interpretation
Many genetic variants remain classified as variants of uncertain significance (VUS), making clinical interpretation difficult.
Diversity in Genomic Databases
Most genomic studies have focused on populations of European ancestry, raising concerns about equity and generalizability in genomic medicine. ([Nature][1])
Data Privacy
Integrating genomic data with clinical records raises important questions about patient privacy and data security.
Infrastructure Costs
Implementing large-scale precision medicine systems requires major investments in sequencing, data storage, and computational infrastructure.
The Future of Precision Medicine
Despite these challenges, the integration of genomics and multi-omics is rapidly reshaping biomedical science. Future healthcare systems may routinely incorporate:
- genome sequencing at birth
- AI-driven clinical decision support
- predictive disease risk modeling
- personalized prevention strategies
Ultimately, the goal is to shift medicine toward P4 healthcare:
- Predictive
- Preventive
- Personalized
- Participatory
By integrating genetic information, biological pathways, environmental exposures, and clinical data, precision medicine promises a deeper understanding of disease and more effective care for patients.
Check out how multi-omics data can be transformed into images for analysis using Artificial Intelligence!