Introduction
Precision medicine has enormous potential, but its misuse or misinterpretation can lead to confusion, missed opportunities, or even patient harm. Understanding these pitfalls is essential for responsible implementation.
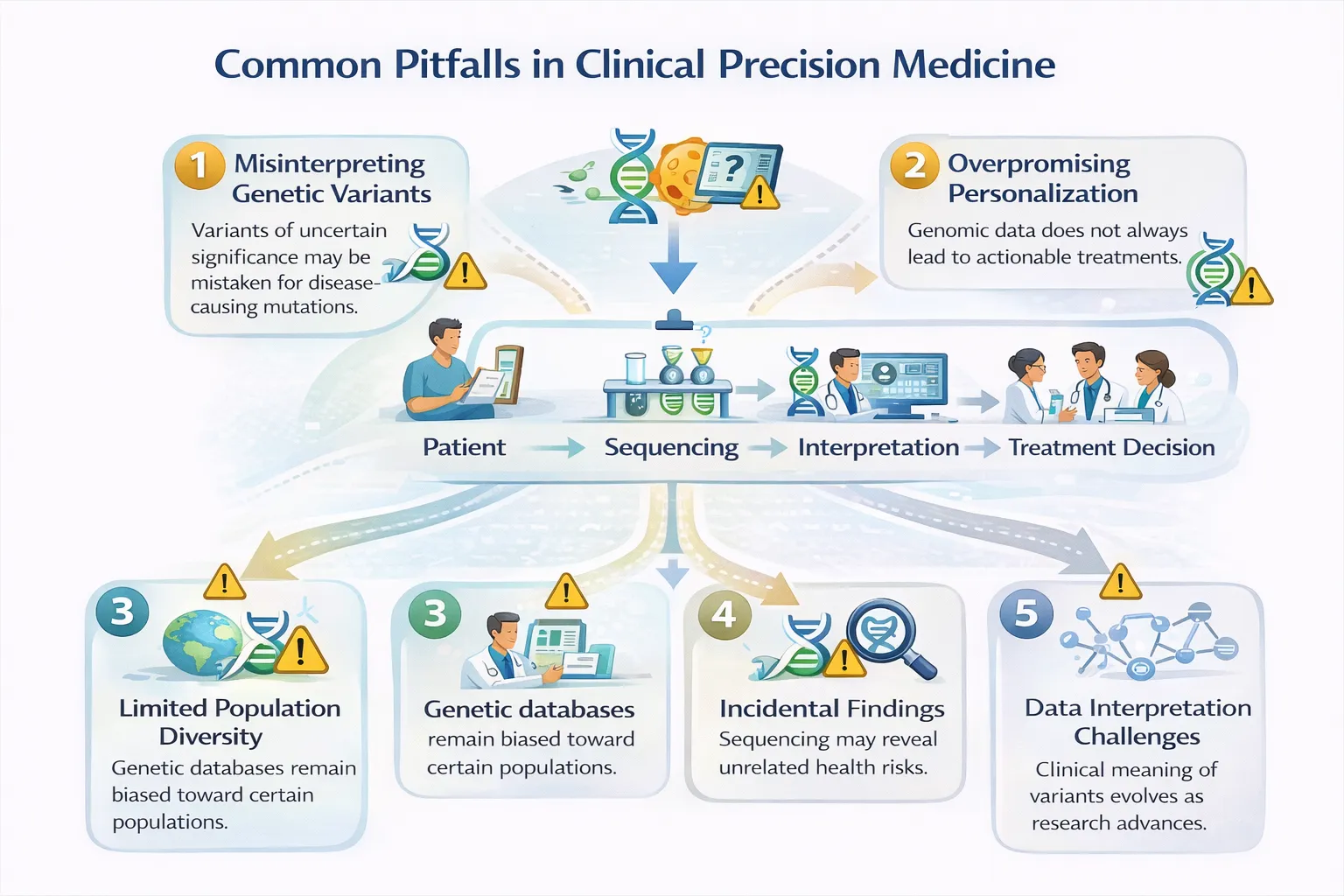
1. Assuming Genetic Results Are Always Actionable
The pitfall There is a widespread assumption that identifying a genetic variant automatically leads to a treatment change.
The reality Most detected variants:
- Have uncertain clinical significance
- Are not linked to approved therapies
- May lack strong evidence in diverse populations
Only a small subset of genomic findings are currently actionable, particularly outside oncology and pharmacogenomics.
Why it matters Overstating actionability can create false hope for patients and inappropriate pressure on clinicians.
2. Confusing Risk with Diagnosis
The pitfall Polygenic risk scores and susceptibility variants are sometimes interpreted as diagnoses.
The reality
- Genomic risk ≠ disease
- Many high-risk individuals never develop illness
- Many patients with disease have average genetic risk
Environmental exposures, lifestyle, and social determinants often outweigh genetic risk.
Clinical consequence Unnecessary anxiety, over-screening, or overtreatment.
3. Ignoring Ancestry and Population Bias
The pitfall Applying genomic results derived primarily from European ancestry populations to everyone.
The reality
- Variant frequencies differ across populations
- Polygenic risk scores perform unevenly
- Some “pathogenic” variants are benign in specific ancestries
Efforts such as the Human Pangenome Reference Consortium aim to correct this, but gaps remain.
Patient impact Higher rates of uncertain or incorrect results in under-represented groups.
4. Over-Interpreting Variants of Uncertain Significance (VUS)
The pitfall Treating VUS findings as clinically meaningful.
The reality A VUS means:
- There is insufficient evidence
- The variant should not guide treatment
- Reclassification may take years—or never occur
Best practice Clinical decisions should be based on phenotype, family history, and validated findings—not VUS alone.
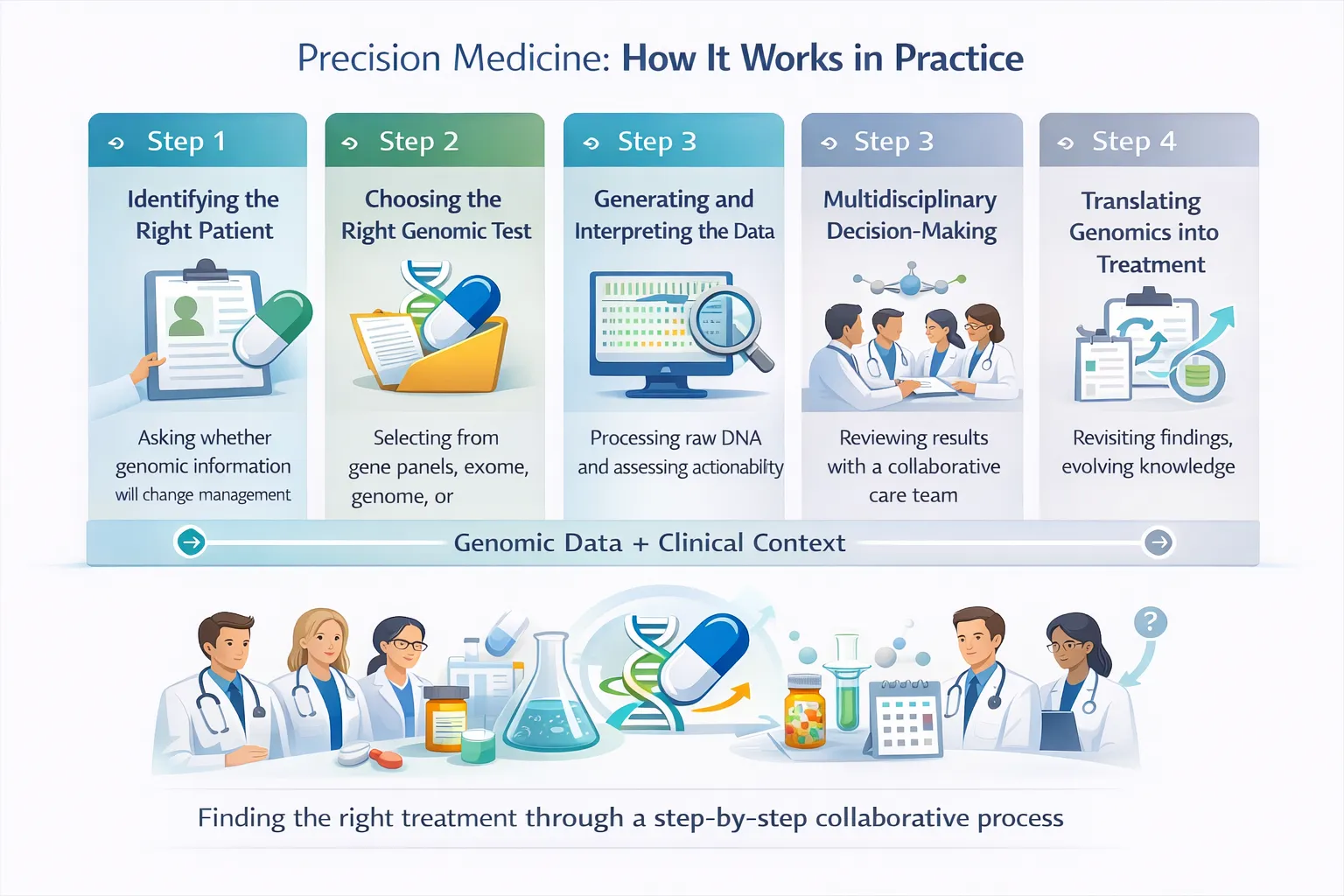
5. Ordering the Wrong Test for the Clinical Question
The pitfall Using broad genomic testing when a targeted test would suffice—or vice versa.
Examples
- Ordering whole genome sequencing for a drug-gene interaction
- Using a small panel when tumour heterogeneity requires broader profiling
Why it matters Inappropriate testing increases cost, complexity, and misinterpretation risk without improving care.
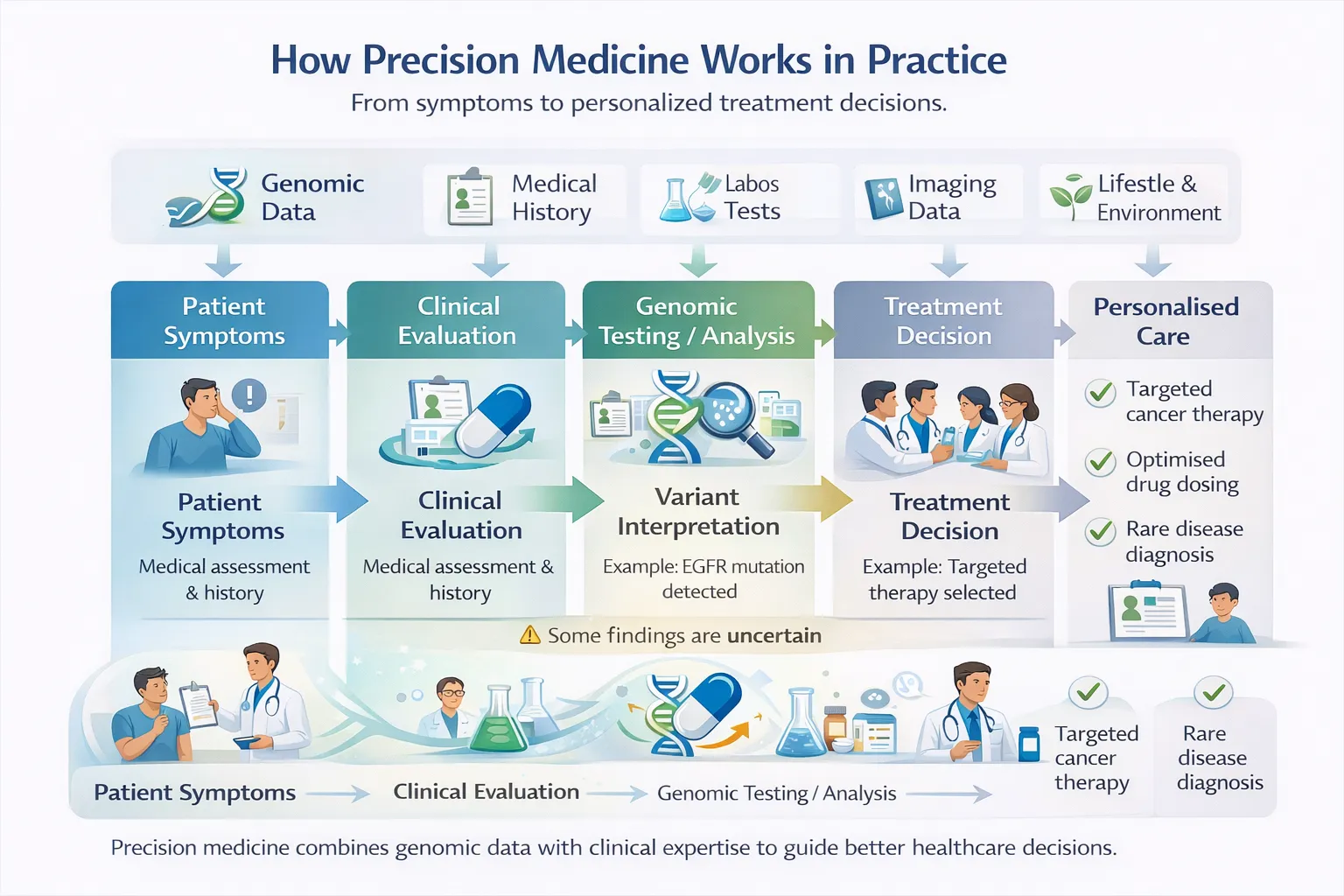
6. Underestimating Interpretation and Counselling Needs
The pitfall Viewing genomic testing as a simple laboratory result.
The reality Genomic results often require:
- Pre-test counselling
- Post-test explanation
- Family cascade testing
- Long-term reinterpretation
Without proper support, patients may misunderstand results or feel abandoned after testing.
7. Treating Precision Medicine as a Replacement for Clinical Judgment
The pitfall Allowing genomic data to override clinical context.
The reality Genomics informs decisions—it does not make them.
Symptoms, examination findings, imaging, laboratory data, and patient values remain essential.
Key principle Precision medicine augments clinical care; it does not automate it.
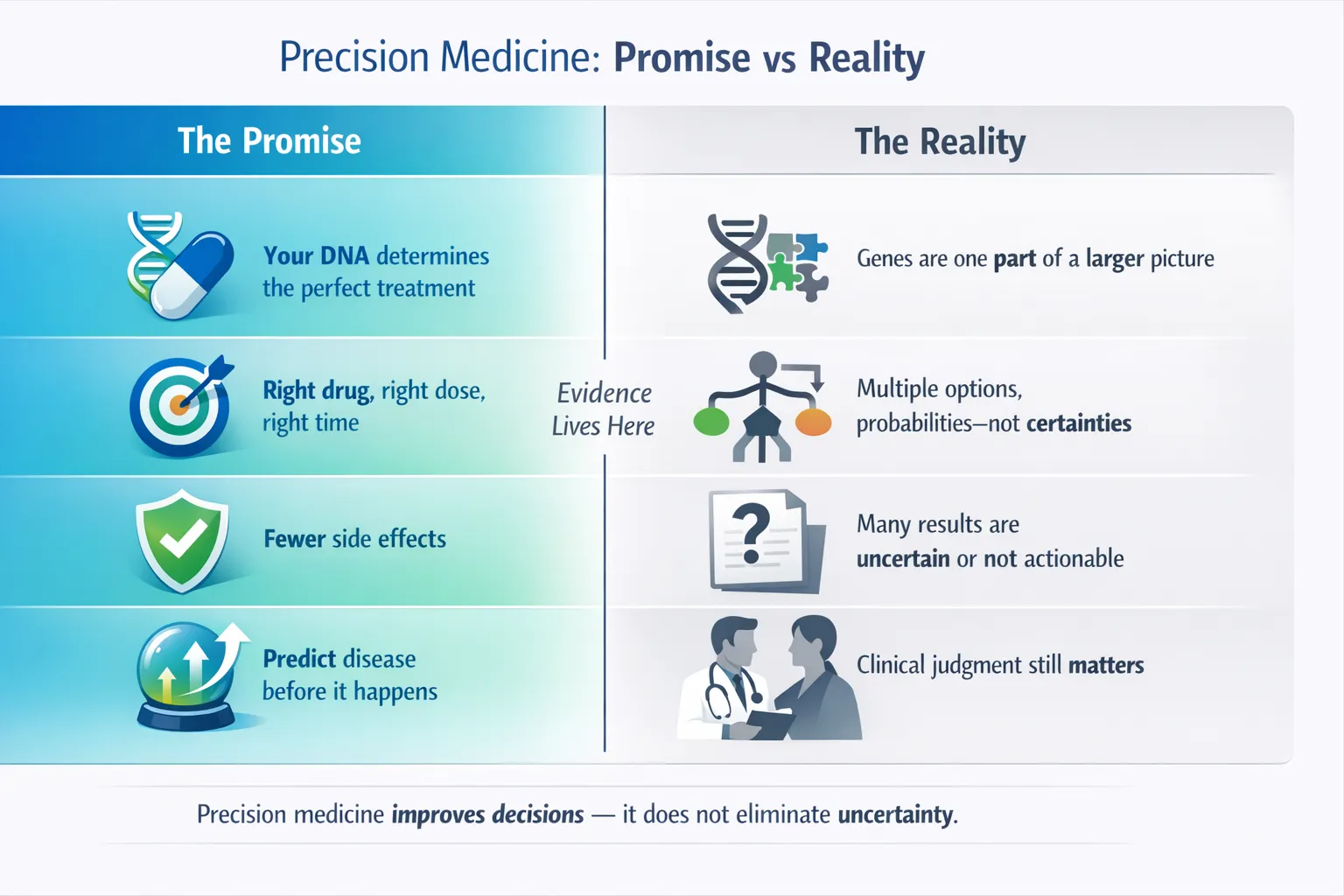
8. Over-Promising Outcomes
The pitfall Presenting precision medicine as guaranteed, curative, or universally beneficial.
The reality
- Many patients receive no actionable result
- Some findings raise more questions than answers
- Benefits may be probabilistic rather than definitive
Transparency protects trust.
What Good Precision Medicine Looks Like
Avoiding these pitfalls requires:
- Clear clinical questions
- Appropriate test selection
- Multidisciplinary interpretation
- Honest communication of uncertainty
- Ongoing education for clinicians and patients
When practiced responsibly, precision medicine improves care—not by eliminating uncertainty, but by making uncertainty more manageable and decisions more informed.