Introduction
How genomic diagnostics moved from niche specialty services to a core component of modern medicine
Clinical genetic testing has undergone a profound transformation over the past two decades. What began as targeted single-gene assays ordered for rare Mendelian disorders has evolved into a broad ecosystem of multi-gene panels, chromosomal assays, exome sequencing, and increasingly genome-scale diagnostics. A recent large-scale analysis of electronic health records (EHRs) from 1.8 million patients at Vanderbilt University Medical Center offers one of the clearest real-world pictures yet of how genetic testing has entered mainstream healthcare. ([Nature][1])
This article explores the major trends identified in that study: expanding test utilization, shifts in technology, rising diagnostic breadth, increasing uncertainty from complex results, and the growing role of genetics across specialties.
Executive Summary
Between 2002 and 2022:
- The number of unique diseases diagnosed with genetic testing each year increased 10-fold (from 51 to 509)
- Recorded clinical genetic tests increased to 104,392 tests across 77,033 patients
- The proportion of patients with documented genetic testing rose from 1.0% to 6.1%
- Molecularly confirmed diagnoses increased substantially
- Multi-gene panels became the dominant testing modality
- Exome/genome sequencing emerged as a high-yield tool
- Variants of uncertain significance (VUSs) increased rapidly
- Genetic diagnoses explained measurable portions of disease burden across the phenome
- Creation of the CGdb, a curated database linking tests, variants, diagnoses, and phenotypes
These findings indicate that clinical genetics is no longer peripheral—it is increasingly embedded within routine patient care.
1. The Rise of Genetic Testing in Real Clinical Practice
The study analyzed two decades of EHR data and found a six-fold increase in the proportion of patients with genetic testing documented in their records. In 2002, only around 1 in 100 patients had evidence of testing. By 2022, that number exceeded 6 in 100. ([Nature][1])
This growth reflects several converging forces:
Falling sequencing costs
The cost of sequencing and genotyping has dropped dramatically, enabling broader access.
Expansion of actionable indications
Testing is now relevant across oncology, cardiology, reproductive medicine, neurology, pediatrics, pharmacogenomics, and rare disease diagnosis.
Better clinician awareness
Genetics has shifted from a specialist discipline to a distributed capability used by many specialties.
Precision medicine adoption
Health systems increasingly recognize genetic data as useful for diagnosis, risk stratification, and treatment planning.
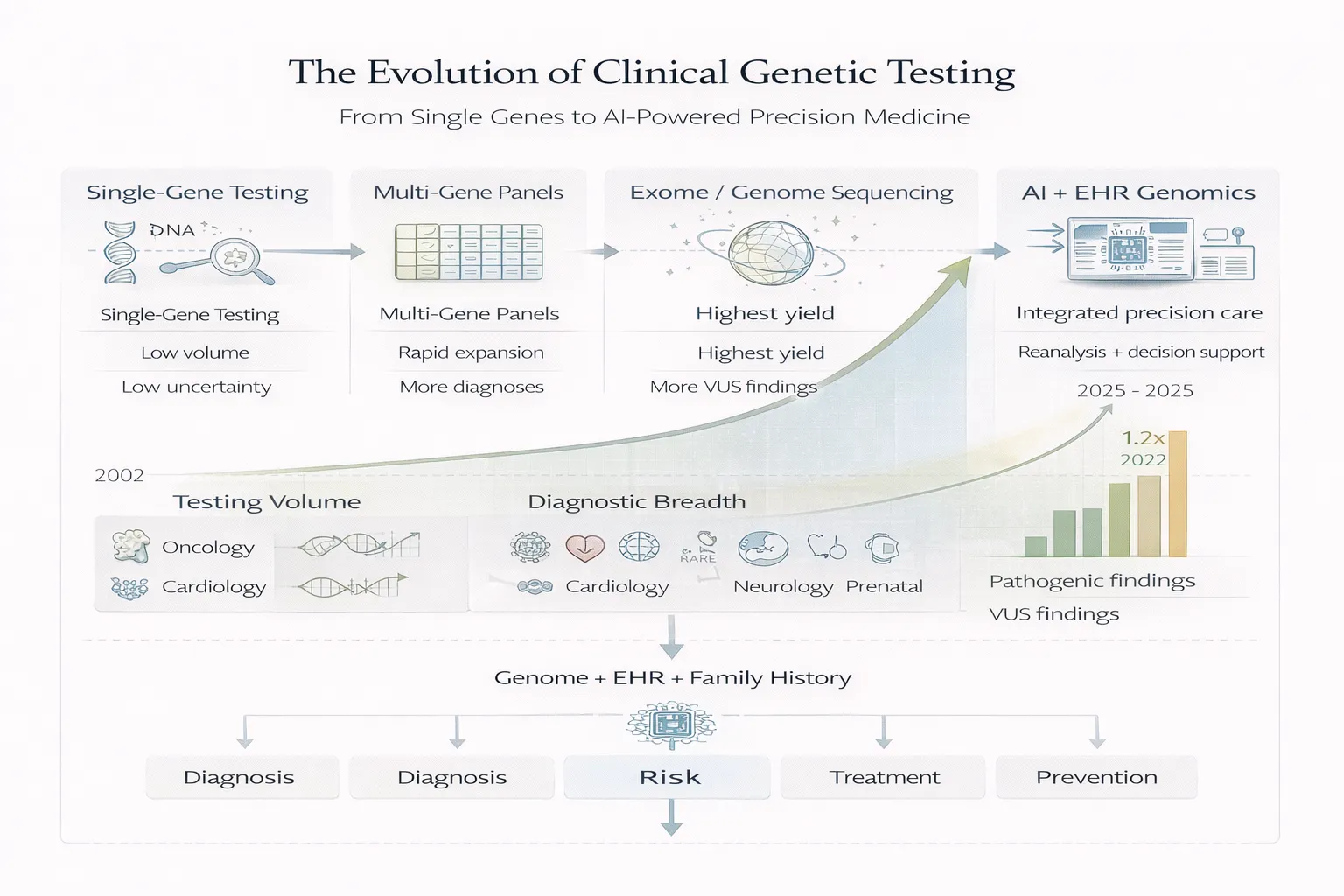
2. Evolution of Testing Modalities
One of the most important findings from the publication is that testing did not simply increase—it became more comprehensive.
Early Era: Single-Gene and Single-Variant Testing
In 2002, the dominant approaches were:
- Single-gene tests
- Single-variant assays
- Targeted mutation detection
These tests were hypothesis-driven: clinicians suspected one disease and tested one gene.
2.1 Middle Era: Cytogenetics and Structural Genomics
Chromosomal microarray (CMA) and karyotyping became standard in developmental delay, congenital anomalies, and reproductive medicine.
2.2 Modern Era: Multi-Gene Panels
By 2015, multi-gene panels overtook single-gene tests and by 2022 represented 46.3% of testing. ([Nature][1])
Panels changed practice because they:
- Test multiple plausible genes simultaneously
- Improve efficiency
- Reduce diagnostic odyssey
- Better handle phenotypic overlap
2.3 Genome-Scale Era: Exome and Genome Sequencing
The first exome sequencing (ES) and gnome sequencing (GS) cases appeared in 2011. By 2022, 349 new ES/GS tests were documented—surpassing karyotypes in that year. ([Nature][1])
These approaches are particularly valuable when phenotype is unclear or genetically heterogeneous.
3. Diagnostic Breadth Expanded Dramatically
As test complexity increased, so did the number of diseases that could be diagnosed.
- Unique diseases diagnosed with genetic testing rose from 51 in 2002 to 509 in 2022
- Multi-gene panels diagnosed 984 unique disorders
- Exome/genome sequencing diagnosed 410 disorders
- 168 conditions were identified only through ES/GS and not by other methods ([Nature][1])
Why this matters
Modern genetics is not only diagnosing more patients—it is diagnosing more different kinds of disease.
This is especially important for:
- Ultra-rare syndromes
- Atypical presentations
- Conditions spanning multiple organ systems
- Previously unsolved pediatric disease
3. The Clinical Genetics Database (CGdb)
One of the most important contributions of the paper is the development of the Clinical Genetics Database (CGdb)—a structured resource created from real-world EHR data to systematically study clinical genetic testing at scale.
What is CGdb?
CGdb is a curated database that integrates:
- Clinical genetic test orders
- Test reports
- Variant classifications
- Diagnoses
- Phenotypic data from EHR records
- Temporal testing trends
- Disease associations across specialties
The database was built from multiple forms of clinical documentation, including structured records and unstructured reports, then harmonized into a research-grade resource.
Why CGdb Matters
Clinical genetics data are often fragmented across:
- PDFs
- scanned reports
- free-text notes
- external laboratory documents
- pathology systems
- specialist clinic correspondence
CGdb addresses this fragmentation by converting heterogeneous clinical genetics information into an analyzable dataset.
Core Components Highlighted in the Publication
Based on the figures and tables in the paper, CGdb captures:
| Component | Description |
|---|---|
| Patients | Individuals with documented genetic testing |
| Tests | Assays across molecular, cytogenetic, and sequencing categories |
| Variants | Pathogenic, likely pathogenic, VUS, benign where available |
| Diagnoses | Genetically linked disorders across specialties |
| Time Trends | Longitudinal changes in utilization and modality |
| Phenotypes | EHR-coded disease manifestations |
| Clinical Context | Ordering specialty and downstream disease burden |
CGdb provides a blueprint for future learning health systems where genomic data can be linked to outcomes, decision support, and AI models.
4. Diagnostic Yield vs Diagnostic Uncertainty
As broader tests became common, clinicians gained more answers—but also more ambiguity.
4.1 Pathogenic Findings
The study identified 28,646 pathogenic variants
4.2 Variants of Uncertain Significance (VUS)
It also identified 25,623 VUSs. By 2018, uncertain findings were accumulating faster than pathogenic ones. By 2022, there were 1.7 VUSs per pathogenic variant. ([Nature][1])
Why VUS Rates Rise
Broad assays interrogate many genes, including genes with:
- Limited evidence
- Rare population frequencies
- Incomplete phenotype data
- Conflicting literature
Clinical Implications
VUS findings can create challenges in:
- Counseling patients
- Cascade family testing
- Surgical decision-making
- Surveillance planning
- Reimbursement decisions
Interpretation infrastructure is now as important as sequencing itself.
5. Which Tests Perform Best?
The publication found that exome/genome sequencing had the highest diagnostic yield: 36.6% positive diagnostic rate
However, it also had one of the highest inconclusive rates 29.8% inconclusive ([Nature][1])
This highlights a key tradeoff:
| Test Type | Breadth | Yield | Uncertainty | Best Use |
|---|---|---|---|---|
| Single-gene | Low | High if phenotype clear | Low | Classic Mendelian disease |
| Panel | Medium | Strong | Moderate | Heterogeneous disorders |
| CMA | Structural | Good in neurodevelopment | Moderate | CNV disorders |
| Exome/Genome | Very high | Highest | High | Complex / unsolved cases |
6. Genetic Testing Across Clinical Specialties
One of the clearest findings of the publication is that clinical genetics has diffused broadly across medicine. Testing is no longer restricted to genetics clinics. This diffusion of genetics into routine specialties is one of the most important structural trends in healthcare.
Oncology
Cancer-related testing represented a major use case.
Included indications:
- hereditary breast/ovarian cancer
- Lynch syndrome
- colorectal cancer predisposition
- endocrine tumor syndromes
- inherited hematologic malignancy risk
Testing helps with:
- prevention
- cascade family testing
- targeted therapies
- surveillance planning
Cardiology
Genetic testing increasingly supports cardiovascular medicine.
Common areas include:
- cardiomyopathy
- inherited arrhythmia syndromes
- sudden cardiac death risk
- aortopathy/connective tissue disease
This enables screening of relatives and earlier intervention.
Neurology
A large and growing area of use.
Includes:
- epilepsy panels
- neuromuscular disease testing
- hereditary neuropathies
- ataxias
- neurodevelopmental disorders
- movement disorders
The publication suggests neurology benefits substantially from multi-gene and exome approaches because of marked genetic heterogeneity.
Pediatrics / Developmental Medicine
One of the strongest clinical domains for testing. Used in:
- congenital anomalies
- developmental delay
- autism spectrum disorder
- metabolic disease
- syndromic presentations
Exome/genome sequencing is especially impactful in pediatric diagnostic odyssey cases.
Reproductive / Obstetrics
Testing includes:
- carrier screening
- prenatal diagnosis
- fetal anomaly workup
- recurrent pregnancy loss investigations
Endocrinology & Metabolic Medicine
Testing supports diagnosis of:
- MODY
- endocrine tumor syndromes
- inherited metabolic disease
- monogenic obesity syndromes
Ophthalmology / ENT / Other Specialties
Genetic testing also contributes to:
- inherited retinal disease
- hearing loss
- connective tissue disorders
- renal genetics
- dermatologic syndromes
The study indicates that genetics is transitioning from a specialty consult service into a cross-specialty infrastructure layer supporting many disciplines.
7. Measuring Clinical Impact: Genetically Attributable Fraction (GAF)
The study introduced a novel metric called the Genetically Attributable Fraction (GAF)—the proportion of observed phenotypes linked to confirmed genetic diagnoses.
For 6.5 million diagnoses in 2022 there was an overall phenome-wide GAF = 0.46%
Certain phenotypes had far higher values:
- Pancreatic insufficiency: 67%
- Chorea: 64%
- Atrial septal defect: 24%
- Microcephaly: 17%
- Paraganglioma: 17%
- Ovarian cancer: 6.8% ([Nature][1])
Interpretation
Some conditions are disproportionately genetic and benefit greatly from routine testing.
This creates opportunities for smarter testing pathways and specialty-specific screening strategies.
8. The Hidden Problem: Poor EHR Integration
One of the paper’s most important operational findings is that many test results remain difficult to use.
The authors found that genetic results were stored across:
- Structured pathology reports
- Templates pasted into notes
- Unstructured free text
- External PDFs
- Scanned reports
Nearly 36.9% of tests appeared only in unstructured clinic notes. ([Nature][1])
Why this matters
If results are not computable, health systems cannot reliably use them for:
- Clinical decision support
- Drug alerts
- Family screening
- Population analytics
- AI model development
- Research recruitment
The bottleneck is increasingly informatics—not sequencing.
9. What Comes Next?
The publication strongly suggests the next phase of clinical genetics will depend on:
- Structured genomic data in EHRs with machine-readable results linked to phenotype.
- Automated reinterpretation and Reclassification of VUS findings over time.
- AI-assisted test selection and smarter decisions about panel vs exome vs genome.
- Longitudinal genomic medicine using inherited risk data throughout life.
- Foundation models combining genomics + EHR with integrated prediction systems for diagnosis, prognosis, and therapy response.
10. Conclusion
Clinical genetic testing has moved from a niche specialty tool to an increasingly mainstream component of medicine. Over two decades, testing volumes increased, technologies broadened, diagnostic yield improved, and genetics spread across specialties.
Yet the next challenge is not merely generating more data. It is making genomic information computable, interpretable, and actionable across every clinical specialty.
The future of genetic testing lies at the intersection of:
- sequencing technology
- expert interpretation
- interoperable health records
- AI-enabled decision support
- precision medicine at scale
Explore related insights
- Integrating Genomics into Multimodal EHR Foundation Models
- Prime Editing: Advances in Human Gene Therapy
- Past, Present, and Future of CRISPR Genome Editing Technologies
- Single-Cell Multi-omics Technologies
- Deep Learning for Multi-Omics Data Integration